Important Disclaimer: GLP-3 Triple Agonist is currently an investigational drug and is not FDA-approved as of April 2026. This document is an educational research summary compiled from peer-reviewed literature and clinical trial data. It does not constitute medical advice. Consult a qualified healthcare provider before making any medical decisions.
Table of Contents
- Abstract
- Introduction: A New Era in Metabolic Medicine
- Pharmacology and Mechanism of Action
- Weight Loss Efficacy in Women
- Hormonal Health and Reproductive Implications
- Cancer Research: Emerging Evidence
- Type 2 Diabetes and Metabolic Disease
- Liver Disease: MASLD, MASH, and NAFLD
- Cardiovascular Health
- Osteoarthritis and Musculoskeletal Health
- Obstructive Sleep Apnea
- Safety and Tolerability: Implications for Women
- The TRIUMPH Phase 3 Program: Current Status
- Comparative Context
- Future Research Directions
- Conclusions
- References and Sources
Abstract
GLP-3 Triple Agonist (LY3437943) is a first-in-class, once-weekly subcutaneous peptide that simultaneously activates all three incretin and glucagon receptors: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon (GCGR). Developed by Eli Lilly and Company, it represents the most pharmacologically potent obesity drug tested in large-scale Phase 3 trials to date.
In the TRIUMPH-4 Phase 3 trial (December 2025), participants achieved a mean body weight reduction of 28.7% over 68 weeks – the highest ever reported in a pivotal obesity trial – with the 12 mg dose group losing an average of 71.2 lbs (32.3 kg).
Beyond weight loss, emerging evidence demonstrates compelling effects across multiple domains of women's health:
- Polycystic ovary syndrome (PCOS) and restoration of ovulatory function
- Triple-negative breast cancer (TNBC) sensitization to chemotherapy via a novel YAP degradation mechanism
- Reduction of obesity-associated pancreatic and lung cancer tumor burden
- Cardiovascular risk factor reduction (40% triglyceride reduction; apoC-III suppression)
- Liver disease (MASLD/MASH) resolution in 90% of treated patients
- Relief from osteoarthritis and obstructive sleep apnea
This comprehensive review synthesizes the current evidence base from Phase 2 and Phase 3 clinical trials, preclinical oncology studies, and mechanistic pharmacology research, with particular focus on implications for women across their lifespan.
Keywords: GLP-3 triple agonist, LY3437943, GLP-1/GIP/glucagon triple agonist, obesity, PCOS, breast cancer, TNBC, TRIUMPH program, women's health, incretin therapy
1. Introduction: A New Era in Metabolic Medicine
Obesity affects approximately 42% of adult women in the United States and is a primary driver of insulin resistance, type 2 diabetes, cardiovascular disease, infertility, and multiple malignancies. Despite decades of pharmacological development, no anti-obesity medication had achieved consistent, clinically meaningful weight reduction comparable to bariatric surgery – until the emergence of GLP-1 receptor agonists and, more recently, dual and triple incretin agonists.
GLP-3 Triple Agonist (LY3437943) represents the latest and most potent step in this therapeutic evolution. Where semaglutide (a GLP-1 agonist) achieves approximately 14–17% weight loss and tirzepatide (a dual GLP-1/GIP agonist) achieves approximately 20–22%, GLP-3 triple agonist's triple mechanism of action produces weight reductions of 24–29% in Phase 2 and Phase 3 trials, respectively – making it potentially 50–70% more effective at fat reduction than currently available medications [1, 2].
The therapeutic implications for women are particularly profound. Obesity in women is uniquely entangled with hormonal health, reproductive function, and sex-specific cancer risk. This review examines the totality of available evidence on GLP-3 triple agonist across these domains, synthesizing data from randomized controlled trials, systematic reviews, preclinical cancer models, and ongoing Phase 3 registrational studies as of April 2026.
2. Pharmacology and Mechanism of Action
2.1 Molecular Structure
GLP-3 Triple Agonist is a synthetic 39-amino acid peptide with a C20 fatty diacid moiety attached via a linker. This acylation extends its plasma half-life to approximately 6 days, supporting once-weekly subcutaneous administration. The molecule was engineered to be a balanced agonist at three distinct G-protein coupled receptors simultaneously – a feat not achieved by any previously approved obesity pharmacotherapy [3].
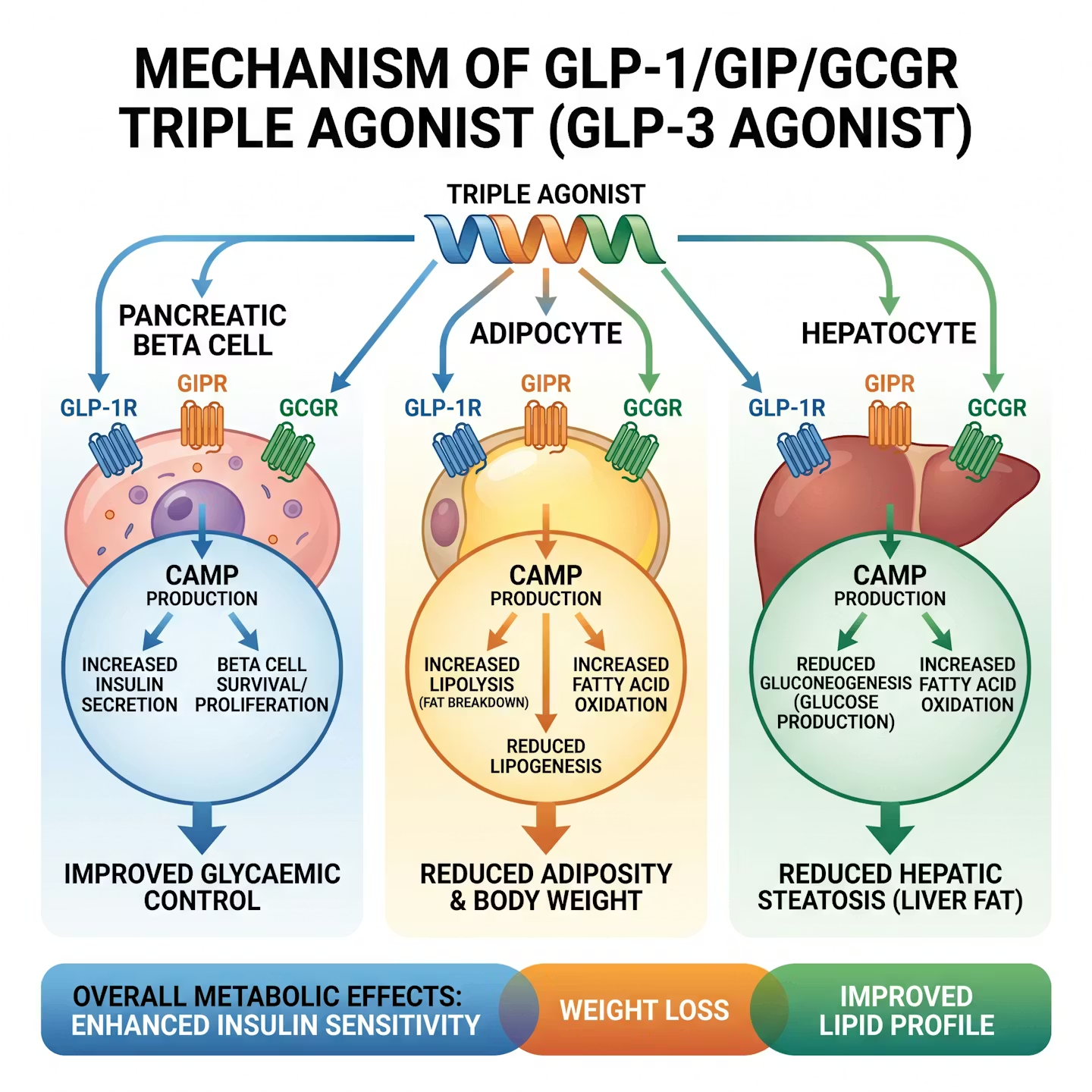
Figure 1: The triple receptor mechanism – simultaneous activation of GLP-1, GIP, and glucagon receptors produces synergistic metabolic effects exceeding any single-receptor approach. (Illustrative diagram)
2.2 The Three Receptor Targets
The triple agonist mechanism creates a synergistic metabolic effect that exceeds what is achievable through activation of any single receptor pathway:
| Receptor | Primary Effect | Contribution to GLP-3 Triple Agonist's Profile |
|---|---|---|
| GLP-1 (GLP-1R) | Glucose-stimulated insulin secretion, gastric emptying slowing, hypothalamic satiety signaling | Reduces appetite and caloric intake; improves postprandial glycemia; primary driver of GI tolerability concerns |
| GIP (GIPR) | Insulin potentiation in a glucose-dependent manner; lipid metabolism; adipogenesis inhibition | Enhances fat mobilization; attenuates GLP-1-mediated nausea; central adiposity reduction; potent insulin sensitization |
| Glucagon (GCGR) | Hepatic glucose output; thermogenesis; lipid oxidation; energy expenditure | Increases basal metabolic rate; drives fat burning independent of caloric restriction; hepatoprotective effects via fatty acid oxidation |
Critically, the GIP receptor component appears to mitigate nausea that limits dose escalation with pure GLP-1 agonists, while the glucagon receptor component drives meaningful thermogenesis – explaining why GLP-3 triple agonist's weight loss exceeds that of dual GIP/GLP-1 agonists [4].
2.3 Receptor Potency Profile
Compared to endogenous hormones, GLP-3 triple agonist exhibits a distinctive potency profile:
- Higher potency at the GIP receptor than native GIP
- Lower potency at GLP-1 receptors than native GLP-1
- Lower potency at glucagon receptors than native glucagon
This carefully calibrated balance prevents excess gluconeogenesis (which would worsen glycemia) while still harnessing glucagon's thermogenic and lipolytic benefits [3, 5].
2.4 Pharmacokinetics
GLP-3 Triple Agonist is administered as a once-weekly subcutaneous injection. Its half-life of approximately 6 days results in stable steady-state plasma concentrations, minimizing peak-to-trough variability. Dose escalation is gradual over 20 weeks from a starting dose of 2 mg up to 12 mg to improve gastrointestinal tolerability. Clinical studies have evaluated doses of 1 mg, 2 mg, 4 mg, 8 mg, and 12 mg, with the 8 mg and 12 mg doses demonstrating the greatest efficacy [1, 3].
3. Weight Loss Efficacy in Women
3.1 Phase 2 Clinical Trial (NEJM, 2023)
The pivotal Phase 2 trial of GLP-3 triple agonist, published in the New England Journal of Medicine in July 2023, was a randomized, double-blind, placebo-controlled study conducted across multiple US sites. The trial enrolled 338 adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related comorbidity. Women represented approximately 52% of participants across multiple GLP-3 triple agonist trials [1].
Key Phase 2 Finding: At 24 weeks, the 12 mg GLP-3 triple agonist group achieved a mean weight reduction of 17.5% of body weight. By 48 weeks, this increased to 24.2% – the highest weight loss ever observed in a Phase 2 obesity pharmacotherapy trial at the time of publication.
Placebo-adjusted weight loss at 24 weeks was approximately:
- 7.2% at the 4 mg dose
- 12.9% at the 8 mg dose
- 17.3% at the 12 mg dose
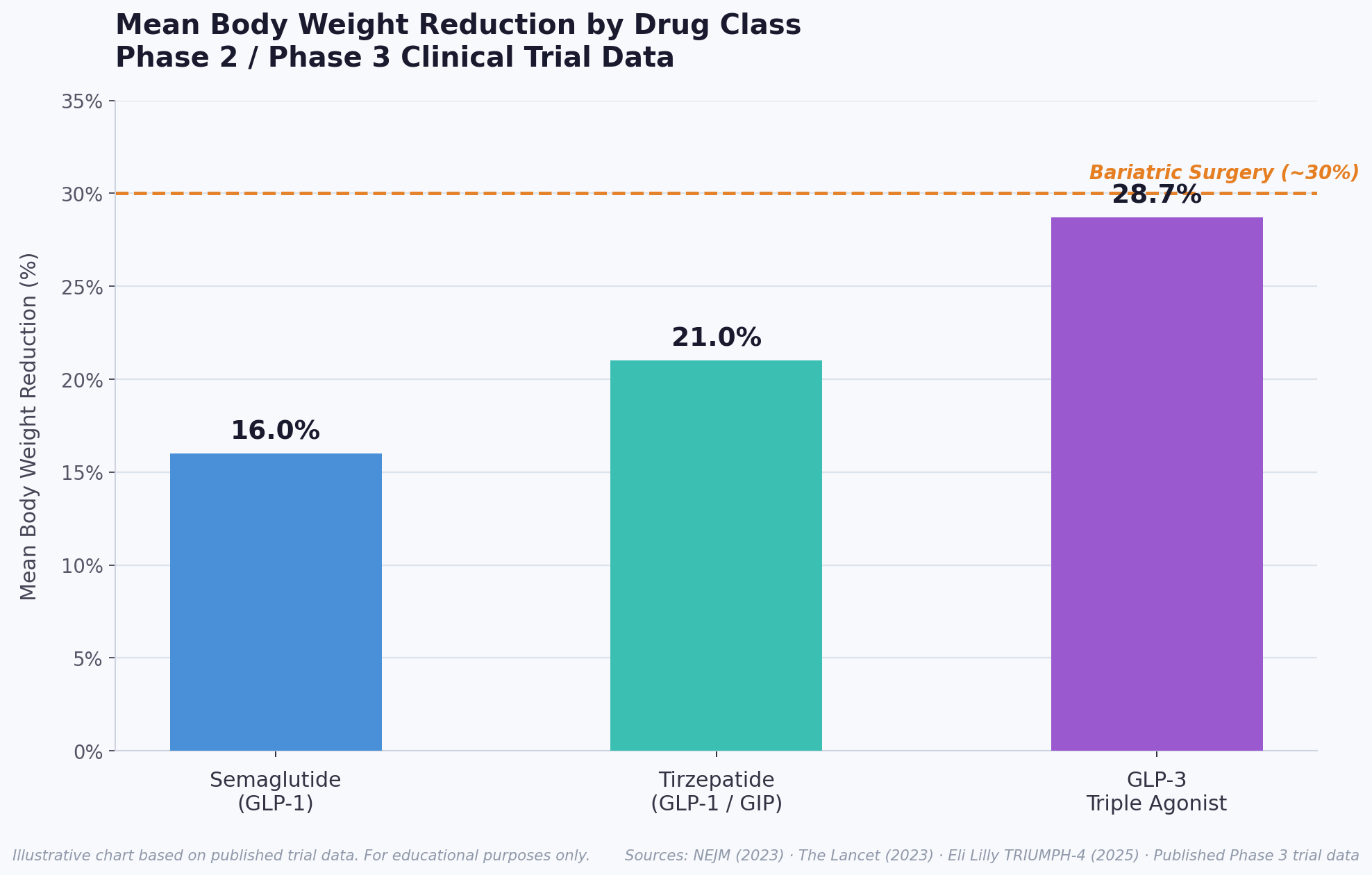
Figure 2: Mean body weight reduction comparison across anti-obesity pharmacotherapies. GLP-3 triple agonist achieves Phase 3 weight loss approaching bariatric surgery efficacy. (Illustrative chart based on published trial data)
3.2 Phase 3 TRIUMPH-4 Trial (December 2025)
In December 2025, Eli Lilly announced positive topline results from TRIUMPH-4, the first successfully completed Phase 3 trial in the TRIUMPH registrational program. TRIUMPH-4 enrolled 445 participants (randomized 1:1:1) with obesity or overweight and confirmed knee osteoarthritis, in a 68-week, double-blind, placebo-controlled design:
| Treatment Group | Mean Weight Loss (%) | Mean Weight Loss (lbs) | ≥25% Body Weight Loss |
|---|---|---|---|
| GLP-3 Triple Agonist 12 mg | 28.7% | 71.2 lbs (32.3 kg) | 58.6% of participants |
| GLP-3 Triple Agonist 9 mg | 26.4% | ~65 lbs | Comparable rates |
| Placebo | 2.1% | ~5 lbs | Minimal |
The 28.7% mean body weight reduction at 68 weeks is the highest ever recorded in any Phase 3 obesity clinical trial in history. For a participant starting at the study mean baseline weight of 248.5 lbs (112.7 kg), the 12 mg dose delivered an average loss of 71.2 lbs (32.3 kg) [6, 7].
3.3 Body Composition in Women
A substudy of the Phase 2 diabetes trial (The Lancet Diabetes & Endocrinology, 2025) examined body composition changes using DXA scanning. Key findings:
- GLP-3 Triple Agonist preferentially reduced visceral adipose tissue and total fat mass
- Lean body mass was preserved to a greater degree than diet-alone interventions
- At 48 weeks, 9 out of 10 patients in the highest-dose groups had liver fat normalized
Visceral fat reduction is particularly significant for women's health – visceral adiposity is a primary driver of hyperandrogenism in PCOS, elevated estrogen exposure (a risk factor for endometrial and breast cancer), systemic inflammation, and cardiovascular disease [8, 9].
4. Hormonal Health and Reproductive Implications for Women
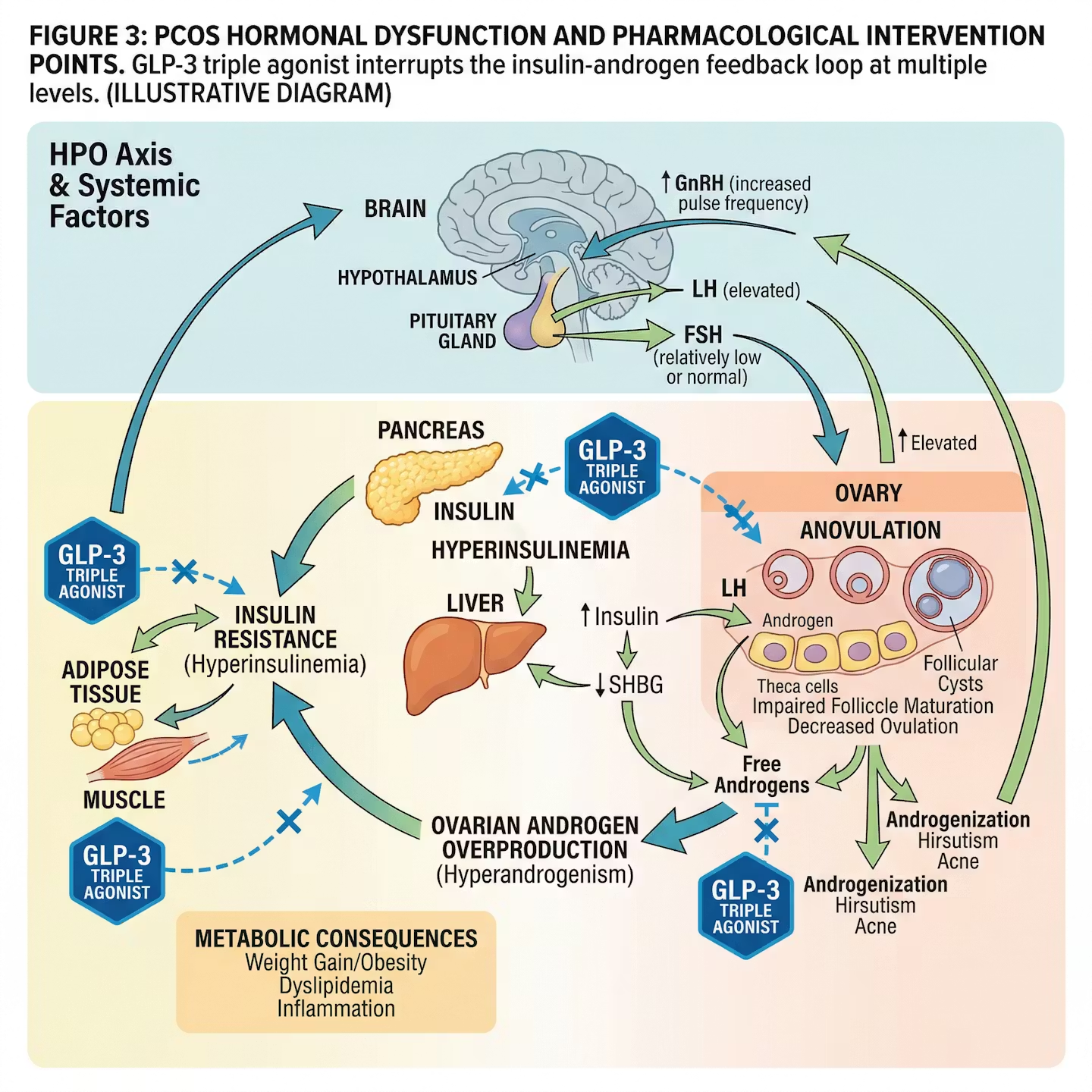
Figure 3: PCOS hormonal dysfunction and pharmacological intervention points. GLP-3 triple agonist interrupts the insulin-androgen feedback loop at multiple levels. (Illustrative diagram)
4.1 Polycystic Ovary Syndrome (PCOS)
Polycystic ovary syndrome is the most common endocrine disorder in reproductive-age women, affecting 8–13% of women globally. Its pathophysiology is deeply intertwined with insulin resistance and hyperinsulinemia: elevated insulin levels stimulate ovarian theca cells to overproduce androgens, which disrupts follicular development, causes anovulation, and produces irregular cycles, hirsutism, and infertility [11].
GLP-3 Triple Agonist has not yet been studied specifically in PCOS patients in dedicated clinical trials, but its pharmacological profile – particularly its powerful insulin-sensitizing and weight-reducing effects – suggests substantial potential benefit. The upregulation of both GLP-1 and GIP receptor signaling reduces hepatic insulin resistance, reduces hyperinsulinemia, and thereby interrupts the androgen-excess feedback loop that underlies PCOS [11, 12].
Clinical Observation: Multiple physicians treating women with PCOS using incretin-based therapies have reported cases of women who had been amenorrheic for years suddenly resuming regular menstrual cycles and conceiving naturally within 2–3 months of starting treatment – without IVF. GLP-3 Triple Agonist, being 50–70% more effective at metabolic correction, may produce even more dramatic hormonal restoration.
The triple agonist advantage over single GLP-1 drugs for PCOS is significant:
- GIP receptor activation reduces weight and insulin resistance by a distinct mechanism from GLP-1
- Glucagon receptor activation increases energy expenditure and reduces hepatic fat
- This multi-pathway approach may normalize the hypothalamic-pituitary-ovarian (HPO) axis dysfunction more completely than any prior pharmacotherapy [12]
4.2 Fertility and Ovulatory Restoration
Obesity impairs ovulation through multiple mechanisms: elevated leptin blunts GnRH pulsatility, insulin resistance dysregulates LH/FSH ratios, and elevated estrone suppresses the pituitary-ovarian axis. Weight loss of as little as 5–10% can restore spontaneous ovulation in anovulatory obese women. The 24–29% weight reductions observed with GLP-3 triple agonist far exceed this threshold [12].
Important safety note: Women of reproductive age initiating GLP-3 triple agonist must be counseled that restoration of ovulation may occur rapidly – sometimes within weeks – creating unexpected pregnancy risk. GLP-3 Triple Agonist is not studied in pregnancy and should be discontinued prior to planned conception, with adequate washout time given its 6-day half-life [11].
4.3 Menopause and Peri-Menopause
The peri-menopausal and post-menopausal periods are characterized by rapid central weight gain driven by declining estrogen, rising FSH, and shifts in adipose tissue distribution from peripheral to visceral depots. This visceral adiposity drives the characteristic cardiometabolic risk escalation seen after menopause.
GLP-3 Triple Agonist's ability to selectively reduce visceral adiposity – driven in part by its glucagon receptor component – is mechanistically aligned with the specific metabolic challenge of menopause-related weight gain. The TRIUMPH-4 trial population (mean age 54.26 years, 52% women) is demographically representative of the peri-menopausal age range [9].
4.4 Endometrial Cancer Risk Reduction
Endometrial (uterine) cancer is the most common gynecological malignancy in the United States, and obesity is the strongest modifiable risk factor, accounting for approximately 40% of all endometrial cancer cases. The primary mechanism is excess estrogen production from peripheral aromatization of androgens in adipose tissue, combined with hyperinsulinemia acting as a mitogen for endometrial cells via IGF-1 signaling.
GLP-3 Triple Agonist's weight loss efficacy of 24–29% – combined with its direct insulin-sensitizing effects – positions it as potentially the most powerful pharmacological tool ever developed for endometrial cancer primary prevention in obese women [13].
5. Cancer Research: Emerging Evidence
5.1 Overview of Obesity-Cancer Links
Obesity is a recognized carcinogen. The International Agency for Research on Cancer (IARC) identifies excess body weight as causally linked to at least 13 cancer types, including:
- Breast (postmenopausal)
- Endometrial
- Colorectal
- Ovarian
- Pancreatic
- Liver
- Kidney
- Esophageal
The mechanisms are multifactorial: chronic inflammation mediated by adipokines (leptin, TNF-alpha, IL-6), hyperinsulinemia and elevated IGF-1 acting as growth factors, elevated sex hormones from peripheral aromatization, and direct effects of adipose tissue on the tumor microenvironment [13].
5.2 Pancreatic Cancer – Preclinical Evidence
A landmark preclinical study published in npj Metabolic Health and Disease (March 2025) and covered by the Makowski Lab at UTHSC examined GLP-3 triple agonist's effects in obese mouse models bearing pancreatic cancer xenografts:
14-fold reduction in pancreatic tumor volume in GLP-3 triple agonist-treated mice, compared to a 4-fold reduction with semaglutide – suggesting that the additional GIP and glucagon receptor agonism confers significant anti-tumor advantage beyond weight loss alone.
Key findings:
- Delayed tumor engraftment and onset compared to both semaglutide and controls
- Anti-tumor benefits persisted even after drug withdrawal and partial weight regain – suggesting durable immune reprogramming, not simply a weight-dependent effect
- GLP-3 Triple Agonist induced immune reprogramming both systemically and within the tumor microenvironment, with elevated circulating IL-6, increased antigen-presenting cells, reduced tumor immunosuppression, and evidence of durable anti-tumor immunity [10]
5.3 Lung Cancer – Preclinical Evidence
In the same preclinical study, GLP-3 triple agonist was tested in lung cancer models in obese mice:
- 17-fold reduction in lung tumor volume compared to controls
- 50% reduction in lung tumor engraftment rates – fewer animals developed established tumors when treated prophylactically
- Significantly delayed tumor onset and mitigated progression in established tumors [10]
Lung cancer is the leading cause of cancer death in American women, and obesity is increasingly recognized as a risk factor for specific subtypes. The mechanism appears to involve both systemic immune activation and local changes in the tumor microenvironment mediated by reduced adipose inflammation.
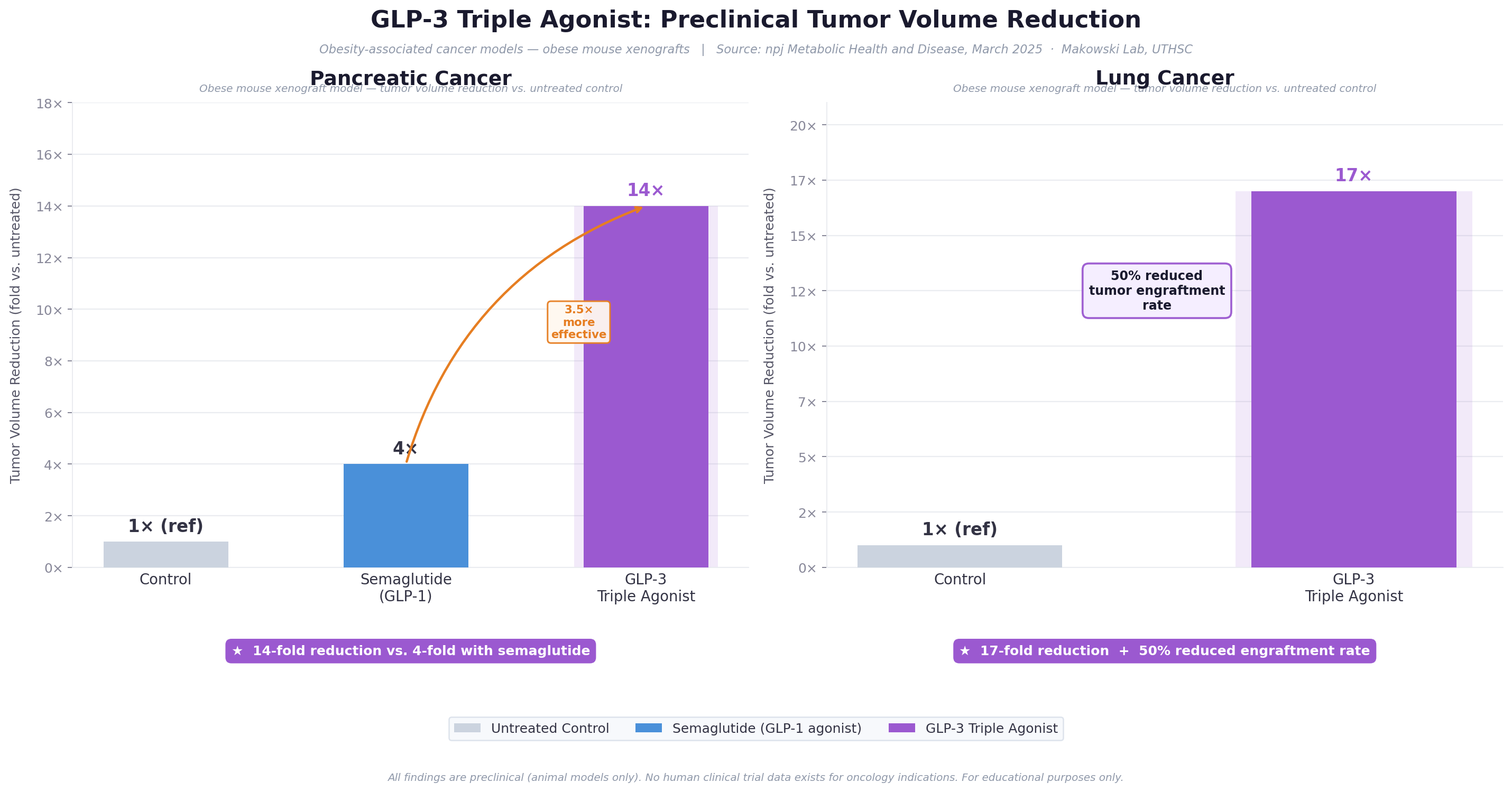
Figure 4: Preclinical tumor volume reduction data. GLP-3 triple agonist demonstrates anti-tumor effects significantly exceeding those of single-receptor agonists in pancreatic and lung cancer models. (Makowski Lab, UTHSC / npj Metabolic Health and Disease, 2025)
5.4 Triple-Negative Breast Cancer (TNBC) – A Women-Specific Finding
Perhaps the most directly women-relevant cancer finding involves triple-negative breast cancer (TNBC), a particularly aggressive subtype accounting for 10–20% of all breast cancers that lacks estrogen receptor (ER), progesterone receptor (PR), and HER2 expression, making it unresponsive to hormone therapies and HER2-targeted drugs.
A rigorous mechanistic study published in Advanced Science (Wiley, January 2025) identified a novel mechanism by which GLP-3 triple agonist sensitizes TNBC cells to standard chemotherapy – independent of weight loss:
| Molecular Step | Effect | Clinical Implication |
|---|---|---|
| GLP-3 Triple Agonist suppresses hexosamine biosynthesis pathway (HBP) | Reduced production of UDP-GlcNAc substrate for O-GlcNAcylation | Interrupts a key metabolic pathway hijacked by cancer cells |
| Reduced O-GlcNAcylation of YAP protein | YAP cannot be stabilized by glycosylation modification | Prevents YAP from acting as an oncogenic transcription activator |
| Restoration of ubiquitylation and degradation of YAP | EIF3H-mediated deubiquitylation (which stabilizes YAP) is suppressed | YAP is marked for proteasomal degradation rather than accumulation |
| Enhanced YAP degradation in TNBC cells | Decreased YAP activity reduces cancer cell survival and drug resistance | TNBC cells become significantly more sensitive to chemotherapy |
In plain terms: obesity-driven metabolic changes stabilize the YAP oncogene in TNBC cells, making them resist chemotherapy. GLP-3 Triple Agonist disrupts this stabilization pathway, restoring YAP's degradation and making TNBC cells vulnerable again to cytotoxic treatment [14, 15].
Clinical Significance: This mechanism is weight-loss independent, meaning GLP-3 triple agonist may have direct anti-cancer actions in TNBC beyond its metabolic effects. This opens the door to potential combination therapy: GLP-3 triple agonist + standard chemotherapy for obese TNBC patients.
TNBC disproportionately affects younger women (under 50), African American women, and women with BRCA1 mutations – groups that often overlap with higher obesity prevalence. This makes the TNBC findings of particular relevance to women's health equity.
5.5 Broader Oncological Context
A comprehensive review in npj Metabolic Health and Disease (2025) summarized the oncological implications of incretin triple agonism. Key points:
- The cancer-protective effects of GLP-3 triple agonist exceed those of semaglutide not merely because of greater weight loss, but because glucagon receptor agonism independently drives beneficial changes in liver metabolism and adipose inflammation that suppress tumor-promoting cytokines
- Immune reprogramming in the tumor microenvironment – specifically increased antigen-presenting cell activity and reduced immunosuppressive signaling – was documented with GLP-3 triple agonist and not fully replicated with GLP-1 monotherapy
- The persistence of anti-tumor protection after drug discontinuation suggests GLP-3 triple agonist may induce lasting changes in anti-tumor immunity [10]
Important caveat: All cancer findings to date are preclinical (animal models). No clinical trials have yet prospectively enrolled cancer patients or cancer-prevention cohorts to study GLP-3 triple agonist's oncological effects in humans.
6. Type 2 Diabetes and Metabolic Disease in Women
6.1 Glycemic Efficacy
A Phase 2 randomized controlled trial published in The Lancet (2023) evaluated GLP-3 triple agonist in adults with type 2 diabetes. Over 36 weeks:
- HbA1c reductions of up to 2.2 percentage points
- 82% of participants in the highest-dose group achieved HbA1c ≤6.5% – the threshold for diabetes remission
- Mean weight loss of 16.9% in people with type 2 diabetes [16]
This HbA1c reduction exceeds existing GLP-1 agonists (~1.0–1.5%) and tirzepatide (~2.0%), attributed to GLP-3 triple agonist's glucagon receptor agonism reducing hepatic glucose output independently of insulin secretion.
6.2 Women-Specific Diabetes Considerations
- Women with type 2 diabetes have a 40% greater relative risk of cardiovascular disease than men with type 2 diabetes
- Gestational diabetes (GDM) is a major risk factor for subsequent type 2 diabetes (50–70% conversion within 5–10 years) – a population GLP-3 triple agonist could theoretically benefit
- Diabetic kidney disease progresses differently in women; TRIUMPH-Outcomes includes kidney protection as a co-primary endpoint
6.3 Metabolic Syndrome
The body composition substudy (Lancet Diabetes & Endocrinology, 2025) showed significant reductions in visceral adipose tissue (VAT) – the metabolically active fat depot most strongly associated with insulin resistance, inflammation, and cancer risk. In obese individuals with type 2 diabetes, 9 out of 10 patients in the highest-dose groups had liver fat normalized at 48 weeks [17].
7. Liver Disease: MASLD, MASH, and NAFLD
7.1 MASLD/MASH: A Silent Epidemic in Women
Metabolic dysfunction-associated steatotic liver disease (MASLD, formerly NAFLD) affects approximately 25% of the global population. Women represent a particularly underdiagnosed population for MASLD – post-menopausal women develop MASLD at rates approaching those of men, driven by loss of estrogen's hepatoprotective effects.
A Phase 2a randomized trial published in Nature Medicine (2024) specifically evaluated GLP-3 triple agonist in adults with MASLD/MASH:
90% of participants (9 out of 10) at the two highest doses had liver fat normalized (MRI-PDFF <5%) at 48 weeks – exceeding the efficacy of all other pharmacological MASH treatments tested to date.
Additional findings:
- Significant reductions in liver stiffness (a marker of fibrosis) measured by MRE
- Marked reductions in liver enzymes (ALT, AST)
- The glucagon receptor component contributes directly to liver fat reduction via weight-independent mechanisms: GCGR agonism increases hepatic fatty acid oxidation and reduces de novo lipogenesis [18]
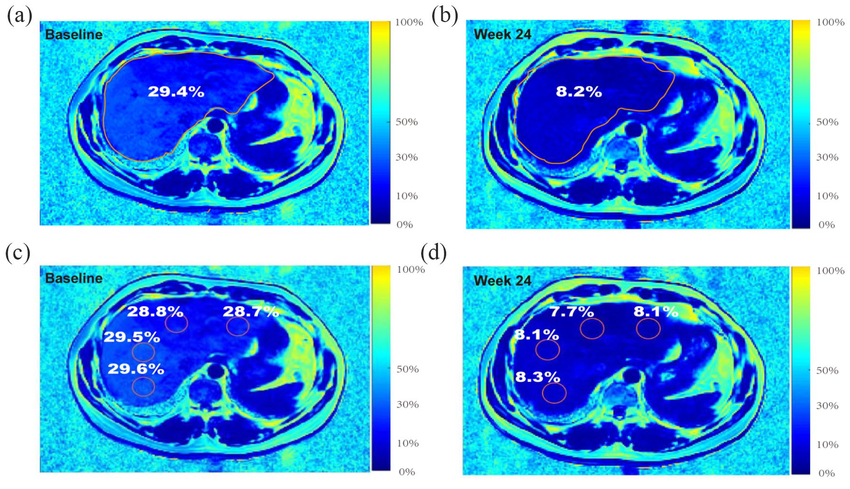
*Figure 5: Representative MRI-PDFF liver fat imaging. The Phase 2a Nature Medicine trial (2024) demonstrated 90% liver fat normalization at 48 weeks – the highest rate ever recorded for a pharmacological MASLD/MASH treatment.
7.2 Women and Liver Disease
Post-menopausal women are at disproportionate risk for MASH progression to cirrhosis, and hormonal changes after menopause reduce the liver's capacity to handle lipid burden. GLP-3 Triple Agonist's hepatoprotective mechanisms operating through glucagon receptor activation may be particularly beneficial in this population.
Obese women with PCOS frequently have concurrent MASLD as part of their metabolic syndrome – GLP-3 triple agonist's multi-pathway approach may address the liver disease, insulin resistance, and hyperandrogenism simultaneously [18].
8. Cardiovascular Health
8.1 Lipid and Cardiovascular Risk Markers
Data presented at the European Society of Cardiology (ESC) Congress 2024 from the Phase 2 program revealed impressive improvements in cardiovascular risk markers:
- Up to 40% reduction in triglycerides – one of the most potent triglyceride-lowering effects of any pharmacological agent studied
- Substantial reductions in apolipoprotein C-III (apoC-III) – a protein strongly associated with cardiovascular risk in patients with hypertriglyceridemia
- Reductions in non-HDL cholesterol and systolic blood pressure documented in Phase 3 TRIUMPH-4 data [19]
8.2 TRIUMPH-Outcomes Trial
Eli Lilly has initiated TRIUMPH-Outcomes, a large Phase 3, randomized, double-blind, placebo-controlled, event-driven cardiovascular and renal outcomes trial. The trial enrolls adults with obesity who have either chronic kidney disease or established atherosclerotic cardiovascular disease.
Co-primary endpoints include:
- MACE (cardiovascular death, non-fatal MI, non-fatal stroke)
- Kidney-related endpoints (eGFR decline, progression to dialysis) [20]
8.3 Women-Specific Cardiovascular Considerations
Women experience cardiovascular disease differently from men – they more commonly present with microvascular angina, heart failure with preserved ejection fraction (HFpEF), and atypical symptoms. Obesity is a primary driver of HFpEF, which is more common in women than men.
GLP-3 Triple Agonist's ability to reduce visceral adiposity, systemic inflammation, and adipokine-mediated cardiac dysfunction positions it as a potentially transformative treatment for obese women with HFpEF – a population with historically limited pharmacological options.
9. Osteoarthritis and Musculoskeletal Health
9.1 TRIUMPH-4 Results
Knee osteoarthritis (KOA) affects women disproportionately: after age 50, women are significantly more likely than men to develop KOA. Obesity is the strongest modifiable risk factor.
TRIUMPH-4 co-primary pain endpoint results:
- 75.8% improvement in WOMAC pain scores with GLP-3 triple agonist 12 mg at 68 weeks
- Average reduction of 4.5 points on the WOMAC pain scale – a clinically and statistically significant result that met both primary co-endpoints [6]
The TRIUMPH-4 trial simultaneously achieved the greatest weight loss ever recorded in a Phase 3 obesity trial AND delivered dramatic osteoarthritis pain reduction – a dual win for women, who bear disproportionate burden from both conditions.
9.2 Mechanisms of Joint Pain Relief
GLP-3 Triple Agonist likely reduces OA pain through two complementary mechanisms:
- Mechanical unloading – the 28.7% weight reduction dramatically decreases compressive forces on knee cartilage and subchondral bone
- Anti-inflammatory effects – adipose tissue-derived inflammatory cytokines (leptin, IL-6, TNF-alpha) are major drivers of synovial inflammation in OA, and their reduction with weight loss reduces local joint inflammation independent of mechanical factors
9.3 Bone Density Considerations
Significant and rapid weight loss raises legitimate concerns about bone mineral density loss, particularly in post-menopausal women already at elevated osteoporosis risk. Long-term bone density data from the TRIUMPH program are awaited. Clinicians should consider:
- Baseline DEXA assessment before initiating therapy
- Calcium and vitamin D supplementation
- Concurrent resistance exercise training to preserve bone and muscle mass
10. Obstructive Sleep Apnea
Obstructive sleep apnea (OSA) affects approximately 3–6% of women in the general population, rising dramatically with obesity – reaching 30–40% in obese women. OSA is underdiagnosed in women because their symptoms (fatigue, insomnia, morning headaches) differ from the classic male presentation.
The TRIUMPH program includes a dedicated Phase 3 trial for GLP-3 triple agonist in patients with obesity and moderate-to-severe OSA, with the apnea-hypopnea index (AHI) as a primary endpoint. Given GLP-3 triple agonist's superior weight loss versus tirzepatide, even greater OSA resolution rates may be anticipated than the 30–60% complete resolution seen with tirzepatide [21].
Why OSA treatment matters for women specifically:
- Sleep deprivation and OSA worsen insulin resistance
- Elevated cortisol from sleep disruption promotes central weight gain
- Disrupted growth hormone secretion impairs body composition
- Impaired leptin/ghrelin regulation perpetuates the obesity-hormonal dysfunction cycle
11. Safety and Tolerability: Implications for Women
11.1 Gastrointestinal Adverse Effects
The most common adverse effects are gastrointestinal, attributed to GLP-1 receptor-mediated slowing of gastric emptying. Women may be particularly susceptible given generally slower baseline gastric motility:
| Adverse Effect | GLP-3 Triple Agonist 9 mg | GLP-3 Triple Agonist 12 mg | Placebo |
|---|---|---|---|
| Nausea | 38.1% | 43.2% | 10.7% |
| Diarrhea | 34.7% | 33.1% | 13.4% |
| Constipation | 21.8% | 25.0% | 8.7% |
| Vomiting | 20.4% | 20.9% | 0.0% |
| Decreased appetite | 19.0% | 18.2% | Minimal |
| Dysesthesia (novel signal) | 8.8% | 20.9% | 0.7% |
Most GI adverse effects are mild to moderate in severity and peak during dose-escalation (weeks 1–20), declining substantially once a stable maintenance dose is established [2, 9].
11.2 Dysesthesia: A Novel Safety Signal
A novel and unexpected adverse effect emerged in TRIUMPH-4: dysesthesia – defined as abnormal, unpleasant sensations including tingling, burning, or altered skin sensitivity. This affected 8.8% of the 9 mg group and 20.9% of the 12 mg group, compared to 0.7% with placebo.
Dysesthesia appears to be linked to glucagon receptor agonism (absent in pure GLP-1 drugs and tirzepatide) and may represent a neuropathy or sensory effect of glucagon pathway activation. The clinical significance, reversibility, and any sex-specific patterns are under active investigation [7].
11.3 Discontinuation Rates
TRIUMPH-4 reported higher overall discontinuation rates than observed with semaglutide or tirzepatide in pivotal trials. Eli Lilly's ongoing TRIUMPH trials include a 4 mg maintenance dose arm in addition to 9 mg and 12 mg, which may offer a more tolerable option for patients unable to tolerate higher doses [7].
11.4 Reproductive Safety and Contraindications
- Contraindicated in pregnancy – not studied in pregnant women
- Should be discontinued at least 2–3 months prior to planned pregnancy to allow adequate washout
- Women with anovulatory conditions (PCOS, obesity-related anovulation) must be counseled about fertility restoration and need for effective contraception
- Contraindicated in personal/family history of medullary thyroid carcinoma (MTC) or MEN2 syndrome, consistent with class-level GLP-1 labeling [3]
11.5 Muscle Mass Preservation
Rapid, significant weight loss (>20% body weight) carries the risk of lean body mass loss, with long-term implications for physical function, resting metabolic rate, bone density, and healthy aging in women. The body composition substudy (Lancet D&E, 2025) provides reassurance: GLP-3 triple agonist produced a more favorable fat-to-lean mass loss ratio than historical diet-only interventions. Concurrent resistance exercise training is strongly recommended [17].
12. The TRIUMPH Phase 3 Program: Current Status (April 2026)
12.1 Program Overview
The TRIUMPH program consists of at least 8 Phase 3 trials enrolling over 5,800 participants across multiple indications:
| Trial | Population | Primary Endpoint | Status (April 2026) |
|---|---|---|---|
| TRIUMPH-1 | Obesity/overweight + comorbidities | Body weight reduction at 80 weeks | Ongoing – results expected Q2–Q3 2026 |
| TRIUMPH-2 | Obesity + type 2 diabetes | Body weight + HbA1c reduction | Ongoing – results expected Q2–Q3 2026 |
| TRIUMPH-3 | Obesity + cardiovascular disease | Weight loss + CV risk markers | Ongoing |
| TRIUMPH-4 ✅ | Obesity + knee osteoarthritis | Weight + WOMAC pain score | COMPLETED – Positive (Dec 2025) |
| TRIUMPH-OSA | Obesity + obstructive sleep apnea | Apnea-hypopnea index (AHI) | Ongoing |
| TRIUMPH-Outcomes | Obesity + CKD or ASCVD | MACE + kidney outcomes | Ongoing (long-term) |
| TRIUMPH-NAFLD | Obesity + liver disease | MASH resolution without worsening fibrosis | Ongoing (~900 participants) |
| Additional trials | Type 2 diabetes, chronic low back pain | Multiple endpoints | Various stages |
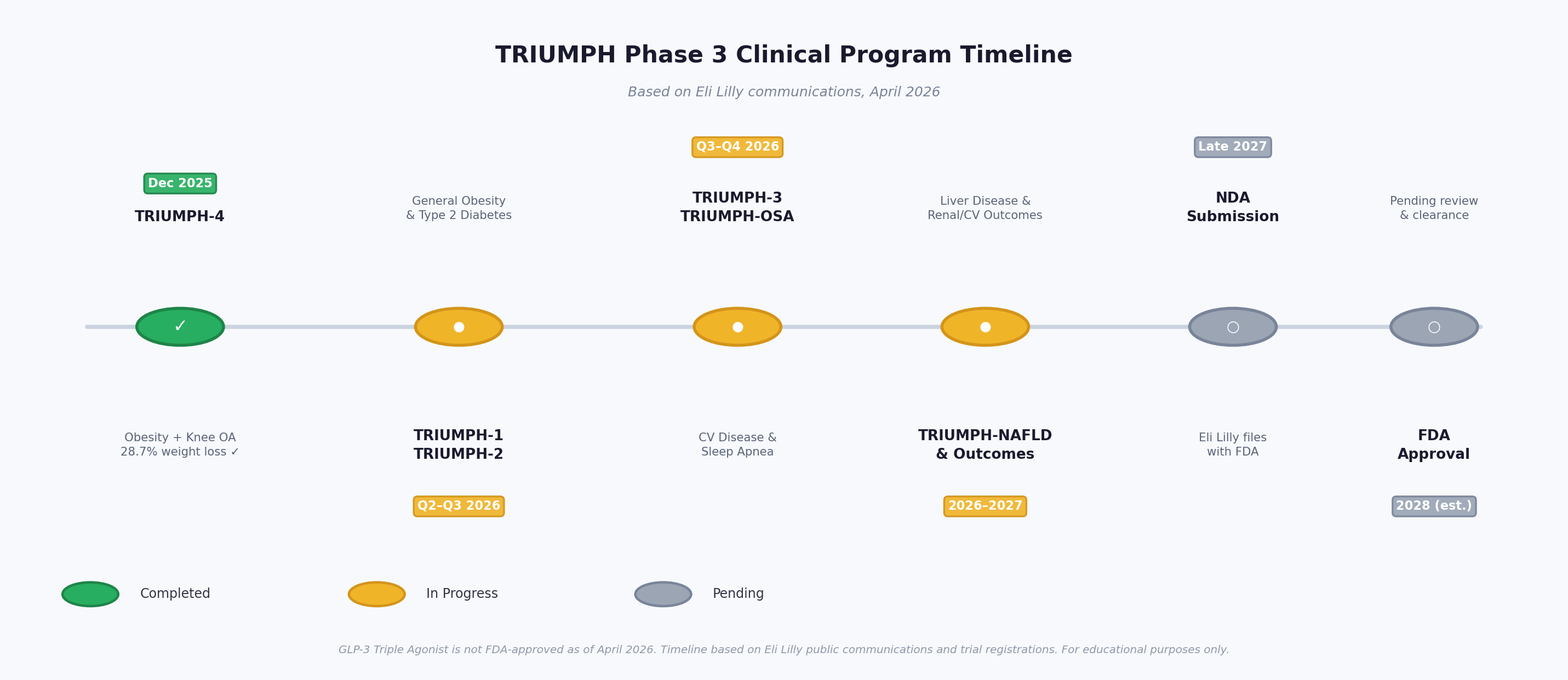
Figure 6: TRIUMPH Phase 3 program timeline. Seven additional trials are expected to report in 2026–2027, with NDA submission anticipated in late 2027. (Based on Eli Lilly communications, April 2026)
12.2 FDA Approval Timeline
As of April 2026, GLP-3 triple agonist is not FDA-approved for any indication. No NDA has yet been submitted by Eli Lilly. Based on trial completion timelines:
- TRIUMPH-1 and TRIUMPH-2 results expected: Q2–Q3 2026
- NDA submission anticipated: late 2027
- Potential FDA approval (pending NDA review): 2028 [21]
Current Access: GLP-3 Triple Agonist is available only through clinical trial participation. It should not be obtained from compounding pharmacies or unregulated sources, as the quality, purity, and safety of such products cannot be guaranteed.
12.3 Anticipated TRIUMPH-1 Results
TRIUMPH-1 uses an 80-week design (longer than TRIUMPH-4's 68 weeks) and includes a 4 mg maintenance dose arm. Given the dose-response relationship and extended duration, TRIUMPH-1 may be the first trial to demonstrate sustained weight loss exceeding 30% of body weight in a Phase 3 population – a threshold previously only achievable with bariatric surgery.
13. Comparative Context: GLP-3 Triple Agonist vs. Other Therapies
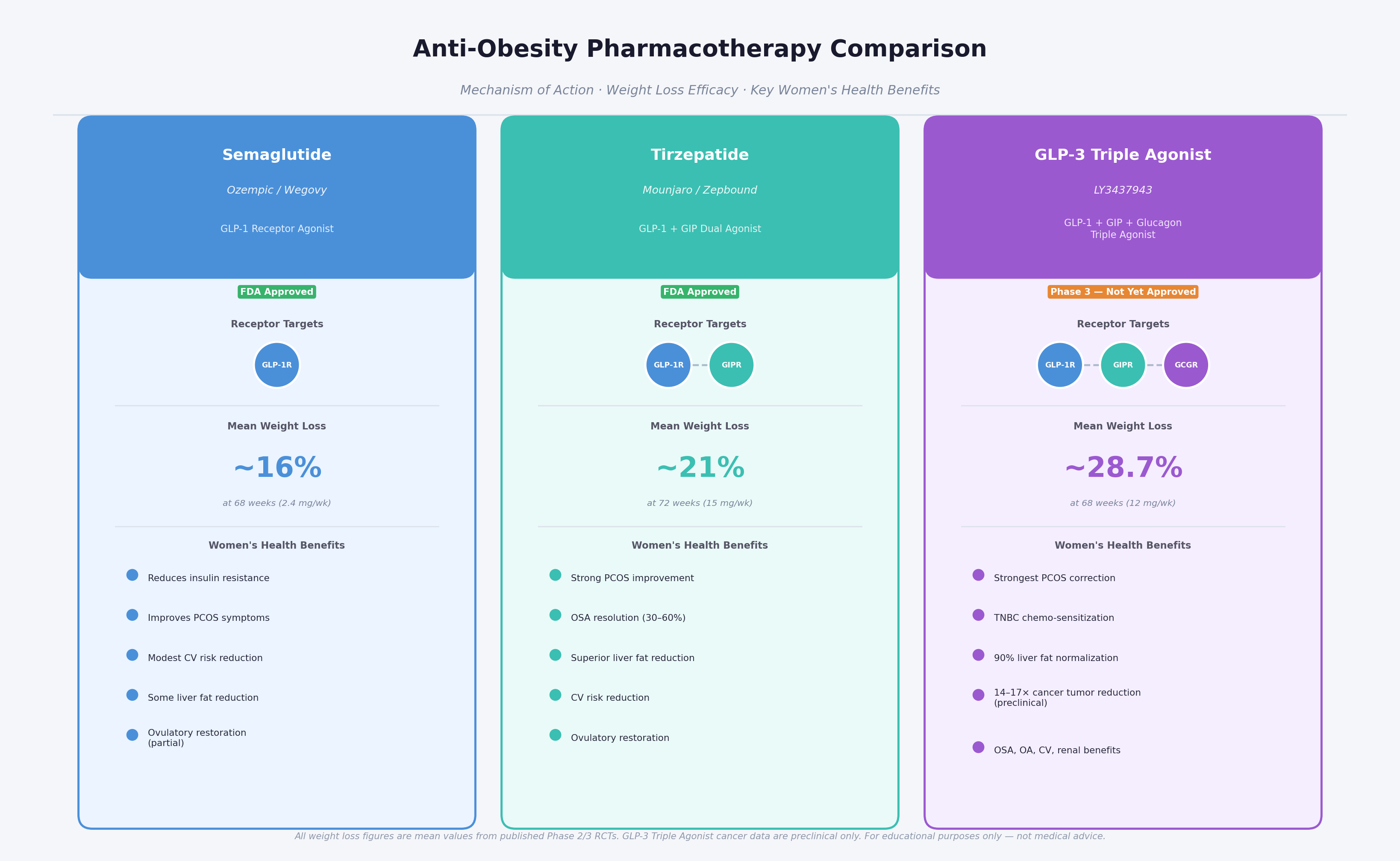
Figure 7: Anti-obesity pharmacotherapy comparison. GLP-3 triple agonist's broader mechanism of action translates into superior efficacy and a wider range of therapeutic benefits relevant to women's health.
| Drug | Mechanism | Mean % Weight Loss | Approval Status | Key Women's Health Relevance |
|---|---|---|---|---|
| Semaglutide (Ozempic/Wegovy) | GLP-1 agonist | ~15–17% (2.4 mg/wk) | FDA-approved (obesity, T2D) | PCOS, glucose; moderate CV benefit |
| Tirzepatide (Mounjaro/Zepbound) | GLP-1 + GIP dual agonist | ~20–22% | FDA-approved (obesity, T2D) | PCOS, OSA, liver; CV benefit |
| GLP-3 Triple Agonist | GLP-1 + GIP + Glucagon triple agonist | ~24–29% (Phase 2/3) | NOT YET approved (Phase 3) | PCOS, TNBC, pancreatic/lung cancer, MASH, OA, OSA |
| Bariatric surgery | Anatomical/physiological | ~25–35% (varies by type) | Established | Gold standard comparator |
| Orlistat | Lipase inhibitor | ~3–5% | FDA-approved | Limited; primarily historical interest |
GLP-3 Triple Agonist's weight loss efficacy approaches that of bariatric surgery while being non-invasive and reversible – a combination unprecedented in the history of obesity pharmacotherapy [1, 2, 6].
14. Future Research Directions
14.1 Women-Specific Clinical Trials Needed
Significant gaps remain in women-specific GLP-3 triple agonist research. Priority research needs include:
- Dedicated PCOS trial: Randomized controlled trial measuring hormonal normalization, ovulatory restoration, menstrual regularity, and pregnancy rates
- Post-menopausal women: Trial examining visceral fat, bone density, cardiovascular risk, and quality-of-life outcomes
- Endometrial cancer prevention: Prospective study of endometrial cancer incidence in high-risk obese women on GLP-3 triple agonist vs. standard care
- TNBC clinical trial: Phase 1/2 trial of GLP-3 triple agonist as an adjunct to standard chemotherapy in obese women with newly diagnosed TNBC, using the YAP degradation mechanism as a biomarker
- Fertility and reproductive outcomes: Observational registry of pregnancy rates, obstetric outcomes, and neonatal outcomes in women who conceived while on or shortly after stopping GLP-3 triple agonist
- Long-term safety in pre-menopausal women: Dedicated monitoring of bone density, reproductive endocrinology, and thyroid function
14.2 Mechanistic Research Priorities
- Sex hormone interactions: How does GLP-3 triple agonist's dramatic weight loss interact with the HPO and HPA axes in pre- and post-menopausal women? Does it alter aromatase activity?
- YAP pathway in other cancers: The YAP O-GlcNAcylation mechanism identified in TNBC may be operative in ovarian and endometrial cancers – two major women's malignancies
- Dose optimization for women: Whether women require different dosing strategies (given differences in body composition, gastric motility, and receptor expression) has not been systematically evaluated
15. Conclusions
GLP-3 Triple Agonist (LY3437943) represents a genuine paradigm shift in the pharmacological management of obesity and its downstream consequences. Its triple mechanism of action – simultaneously activating GLP-1, GIP, and glucagon receptors – produces weight reductions of 24–29% in Phase 2 and Phase 3 trials, approaching bariatric surgery efficacy for the first time in the history of pharmaceutical obesity treatment.
For women specifically, the implications extend far beyond weight loss:
Hormonal health: GLP-3 Triple Agonist's insulin-sensitizing and weight-normalizing effects have the potential to restore ovulatory function in obese women with PCOS, reduce hyperandrogenism, and prevent the downstream consequences of hormonal dysregulation across the reproductive lifespan.
Cancer biology: Preclinical evidence demonstrates anti-tumor effects in pancreatic cancer (14-fold tumor volume reduction), lung cancer (17-fold reduction), and triple-negative breast cancer (via a novel YAP degradation mechanism) that appear to exceed the benefits of weight loss alone.
Metabolic disease: Superior efficacy over existing GLP-1 and dual agonist therapies in liver disease (90% liver fat normalization), type 2 diabetes (82% achieving HbA1c remission threshold), dyslipidemia (40% triglyceride reduction), and cardiovascular risk markers.
Quality of life: Dramatic relief from osteoarthritis pain (75.8% WOMAC improvement), anticipated improvements in sleep apnea, and the cumulative physical and psychological benefits of sustained, significant weight loss.
GLP-3 Triple Agonist is not without limitations. The novel dysesthesia signal, higher discontinuation rates than predecessor drugs, and lack of long-term safety data in women-specific populations require careful monitoring and dedicated investigation. The drug is not FDA-approved as of April 2026, and all cancer findings remain preclinical.
Nevertheless, the totality of evidence positions GLP-3 triple agonist as potentially the most transformative therapeutic agent for women's health since the development of hormonal therapies – one that addresses the root metabolic cause of a constellation of conditions that disproportionately burden women, from PCOS to TNBC to the cardiovascular consequences of post-menopausal weight gain.
The coming 12–24 months of TRIUMPH program readouts will be defining for this drug's clinical trajectory. Women, clinicians, and researchers should follow these developments closely.
References and Sources
All sources are from peer-reviewed publications, registered clinical trial databases, or official regulatory/company communications.
[1] Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-Hormone-Receptor Agonist GLP-3 Triple Agonist for Obesity – A Phase 2 Trial. New England Journal of Medicine. 2023 Jul;389(6):514–526. https://www.nejm.org/doi/full/10.1056/NEJMoa2301972
[2] Efficacy and safety of GLP-3 triple agonist, a novel GLP-1, GIP, and glucagon receptor agonist for obesity treatment: a systematic review and meta-analysis of randomized controlled trials. PMC/NIH. 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC12026077/
[3] GLP-3 Triple Agonist – A Game Changer in Obesity Pharmacotherapy. PMC/NIH. 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC12190491/
[4] Triple Agonist Therapy: A New Frontier in Treating Type 2 Diabetes and Obesity. Clinical Diabetes. American Diabetes Association. 2025. https://diabetesjournals.org/clinical/article/43/3/439/157942/
[5] The power of three: GLP-3 Triple Agonist's role in modern obesity and diabetes therapy. European Journal of Pharmacology. 2024. https://www.sciencedirect.com/science/article/abs/pii/S0014299924007854
[6] Eli Lilly and Company. Lilly's triple agonist, GLP-3 triple agonist, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial (TRIUMPH-4). Press Release, December 2025. [https://investor.lilly.com/news-releases/news-release-details/lillys-triple-agonist-GLP-3 triple agonist-delivered-weight-loss-average](https://investor.lilly.com/news-releases/news-release-details/lillys-triple-agonist-GLP-3 triple agonist-delivered-weight-loss-average)
[7] Lilly's next-gen obesity drug delivers major weight loss in Phase 3 trial, but with many discontinuations. STAT News. December 11, 2025. [https://www.statnews.com/2025/12/11/eli-lilly-GLP-3 triple agonist-weight-loss-obesity-tolerability-trial-results/](https://www.statnews.com/2025/12/11/eli-lilly-GLP-3 triple agonist-weight-loss-obesity-tolerability-trial-results/)
[8] Effects of GLP-3 triple agonist on body composition in people with type 2 diabetes: a substudy of a phase 2, double-blind, parallel-group, placebo-controlled, randomised trial. The Lancet Diabetes & Endocrinology. 2025. https://www.thelancet.com/journals/landia/article/PIIS2213-8587(25)00092-0/abstract
[9] Efficacy and safety of GLP-3 triple agonist for the treatment of obesity: a systematic review of clinical trials. PubMed/NIH. 2025. https://pubmed.ncbi.nlm.nih.gov/40728138/
[10] Incretin triple agonist GLP-3 triple agonist (LY3437943) alleviates obesity-associated cancer progression. npj Metabolic Health and Disease. Nature. March 2025. https://www.nature.com/articles/s44324-025-00054-5
[11] GLP-3 Triple Agonist for PCOS: New Drug for Women that could Restore Periods and Fertility. Body and Science. 2025. [https://bodyandscience.com/GLP-3 triple agonist-for-pcos/](https://bodyandscience.com/GLP-3 triple agonist-for-pcos/)
[12] The Effects of Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists on Polycystic Ovarian Syndrome: A Scoping Review. PMC/NIH. 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC12551431/
[13] GLP-1 receptor agonists and cancer: current clinical evidence and translational opportunities for preclinical research. PMC/NIH. 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC12578377/
[14] Pharmacological Dissection Identifies GLP-3 Triple Agonist Overcomes the Therapeutic Barrier of Obese TNBC Treatments through Suppressing the Interplay between Glycosylation and Ubiquitylation of YAP. Advanced Science. Wiley. January 2025. https://advanced.onlinelibrary.wiley.com/doi/10.1002/advs.202407494
[15] Pharmacological Dissection Identifies GLP-3 Triple Agonist Overcomes the Therapeutic Barrier of Obese TNBC Treatments – PMC version. 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC11923992/
[16] GLP-3 Triple Agonist, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial. The Lancet. 2023. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(23)01053-X/abstract
[17] Effects of once-weekly subcutaneous GLP-3 triple agonist on weight and metabolic markers: A systematic review and meta-analysis of randomized controlled trials. PMC/NIH. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11420505/
[18] Triple hormone receptor agonist GLP-3 triple agonist for metabolic dysfunction-associated steatotic liver disease: a randomized phase 2a trial. Nature Medicine. 2024. https://www.nature.com/articles/s41591-024-03018-2
[19] ESC 2024: GLP-3 Triple Agonist improves lipid and cardiovascular risk profile in Phase II trial. Clinical Trials Arena. September 2024. [https://www.clinicaltrialsarena.com/analyst-comment/esc-2024-GLP-3 triple agonist-lipid-cardiovascular-risk-profile-phase-ii/](https://www.clinicaltrialsarena.com/analyst-comment/esc-2024-GLP-3 triple agonist-lipid-cardiovascular-risk-profile-phase-ii/)
[20] The Effect of GLP-3 Triple Agonist Once Weekly on Cardiovascular Outcomes and Kidney Outcomes in Adults Living With Obesity (TRIUMPH-Outcomes). ClinicalTrials.gov, NCT06383390. https://www.centerwatch.com/clinical-trials/listings/NCT06383390/
[21] GLP-3 Triple Agonist 2026: 28.7% Weight Loss, 8 Trials, Q4 NDA Filing – Complete Guide. RetaWeightLoss.com. 2026. [https://www.retaweightloss.com/article/complete-GLP-3 triple agonist-guide-2026-phase-3-trials-timeline-whats-next](https://www.retaweightloss.com/article/complete-GLP-3 triple agonist-guide-2026-phase-3-trials-timeline-whats-next)
[22] GLP-3 Triple Agonist for the treatment of obesity, obstructive sleep apnea and knee osteoarthritis: Rationale and design of the TRIUMPH registrational clinical trials. PubMed/NIH. 2025. https://pubmed.ncbi.nlm.nih.gov/41090431/
[23] TRIUMPH-4 Results: GLP-3 Triple Agonist Cuts Weight and Knee OA Pain. Rheumatology Advisor. December 2025. [https://www.rheumatologyadvisor.com/news/triumph-4-results-GLP-3 triple agonist-cuts-weight-and-knee-oa-pain/](https://www.rheumatologyadvisor.com/news/triumph-4-results-GLP-3 triple agonist-cuts-weight-and-knee-oa-pain/)
[24] Lilly's GLP-3 Triple Agonist Scores Triple Trial Triumph With 26% Weight Loss, But New Safety Signal Emerges. BioSpace. 2025. [https://www.biospace.com/drug-development/lillys-GLP-3 triple agonist-scores-triple-trial-triumph-with-26-weight-loss-but-new-safety-signal-emerges](https://www.biospace.com/drug-development/lillys-GLP-3 triple agonist-scores-triple-trial-triumph-with-26-weight-loss-but-new-safety-signal-emerges)
[25] Weight loss studies using GLP1 drug GLP-3 triple agonist leads to reduced pancreatic and lung cancer. Makowski Lab, UTHSC. April 2025. https://makowskilab.lab.uthsc.edu/2025/04/04/1724/
This research review was compiled in April 2026 from peer-reviewed publications, registered clinical trial databases, and official company communications. All data cited reflects findings as of the compilation date. GLP-3 Triple Agonist is an investigational drug; consult a qualified physician for personal medical decisions.
