Scope & Disclosure: This article synthesizes findings from peer-reviewed journals, preprint repositories, and registered clinical investigations current through April 2026. Sources are cited inline with footnote markers and compiled in full at the document's end. GHK-Cu remains classified as a research compound in most regulatory jurisdictions. This document is an academic synthesis and does not constitute clinical guidance.
Table of Contents
- Discovery, Chemistry, and Endogenous Biology
- Mechanisms of Action: A Multi-Level Analysis
- The Genomic Scope: 4,000+ Genes and Why That Matters
- Age-Dependent Plasma Decline and Its Convergence with Female Hormonal Biology
- Dermal Biology: Collagen, Elastin, and Extracellular Matrix Research
- Hair Follicle Biology and Alopecia Research in Women
- Neuroprotection, Cognitive Aging, and Alzheimer's Disease Research
- Wound Healing: Cellular Mechanisms and Preclinical-to-Clinical Translation
- Pulmonary Biology: COPD, Silicosis, and Acute Lung Injury
- Bone Turnover, Menopause, and Skeletal Biology
- Reproductive Biology: Endometrial, Ovarian, and Fertility Research
- Cardiovascular Biology and Fibrinogen Suppression
- Gastrointestinal and Hepatic Research
- Cancer Biology: Tumor Suppressor Gene Modulation and Apoptosis Research
- Epigenetics, DNA Repair, Stem Cell Biology, and Aging Clocks
- Bioavailability, Pharmacokinetics, and Delivery System Research
- Safety Pharmacology and Toxicology
- Age-Stratified Biology: What Research Reveals About GHK-Cu Across Women's Life Stages
- Research Gaps and Future Directions
- Complete Source Index
1. Discovery, Chemistry, and Endogenous Biology
1.1 Historical Discovery
GHK was first isolated in 1973 by biochemist Loren Pickart while investigating why young human albumin restored aged liver tissue to functional youth. Through fractionation of human plasma, Pickart identified a small tripeptide — glycyl-L-histidyl-L-lysine — as the active component responsible for the tissue-restoration signal. This discovery positioned GHK not merely as a structural peptide but as a biological age signal: a molecule whose presence communicates tissue integrity and whose absence signals degeneration. 1
The copper-chelated form, GHK-Cu, was subsequently characterized as the physiologically relevant active complex. In biological fluids, GHK exists primarily bound to Cu²⁺, and virtually all documented regenerative and gene-regulatory effects require this chelation. Studies stripping copper from the complex confirm that apo-GHK (without copper) produces substantially attenuated or absent effects in collagen synthesis, antioxidant enzyme activation, and gene modulation assays. 2
1.2 Molecular Chemistry
GHK-Cu is a low-molecular-weight tripeptide (MW ~340 Da without copper; ~404 Da as the copper complex) with the amino acid sequence Gly-His-Lys. The histidine residue provides the primary copper-binding site via its imidazole nitrogen, with additional coordination from the glycine amino terminus and a water molecule completing the square-planar coordination geometry of Cu²⁺. 3
The copper binding affinity (pKa ≈ 16.4) is extraordinary — approximately 10 million times higher than most other tripeptides and nearly equivalent to serum albumin (pKa ≈ 16.2), the dominant copper carrier protein in plasma. This high affinity enables GHK to effectively compete with albumin for copper at sites of tissue damage, delivering Cu²⁺ precisely where regenerative enzymatic activity is required. 2
Critically, chelated copper in GHK-Cu does not behave as free ionic Cu²⁺. Unbound copper drives Fenton-like reactions generating hydroxyl radicals (·OH) from hydrogen peroxide — a major mechanism of oxidative cellular damage. In GHK-Cu, copper is sequestered within the coordination complex, circumventing free radical generation while still enabling delivery to copper-dependent enzymes. This is why GHK-Cu exerts antioxidant effects despite carrying a pro-oxidant metal ion. 3
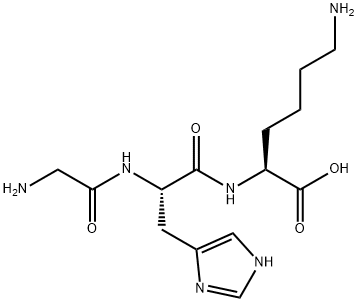
Figure 1: 2D molecular structure of GHK-Cu (PubChem CID 133697840, C₂₈H₄₈CuN₁₂O₈). The square-planar Cu²⁺ coordination is formed by the imidazole nitrogen of histidine, the α-amino nitrogen of glycine, and the deprotonated amide nitrogen of the Gly–His peptide bond. Source: PubChem / NCBI, NIH
1.3 Endogenous Distribution and Function
GHK-Cu is found in:
- Blood plasma — primary reservoir and systemic transport vehicle
- Urine — elimination product and possible local renal signaling
- Saliva — role in oral mucosal repair and gingival tissue maintenance proposed but not fully characterized
- Cerebrospinal fluid (CSF) — detection in CSF implies either central synthesis or blood-brain barrier (BBB) permeability for GHK, with significant implications for neurological research
The peptide's endogenous function is understood as part of a damage-response signaling system. Following tissue injury, matrix metalloproteinases (MMPs) degrade collagen; one product of procollagen degradation is the GHK sequence, which is released locally and enters circulation. Plasma GHK levels rise transiently post-injury, recruiting fibroblasts, promoting angiogenesis, and signaling immune modulation. As repair concludes, levels normalize. This cyclical pattern means GHK functions as a biological wound closure verification signal — its sustained absence, as observed in aging, correlates with progressive failure of tissue maintenance programs. 1
2. Mechanisms of Action: A Multi-Level Analysis
GHK-Cu does not operate through a single receptor-ligand mechanism. Its biological activity is distributed across at least four distinct mechanistic levels: copper delivery, direct enzymatic activation, receptor-mediated transcription factor modulation, and broad epigenetic reprogramming. Understanding why GHK-Cu has such diverse effects requires examining all four levels.
2.1 Copper Chaperoning and Metalloenzyme Activation
Copper is an essential cofactor for over 30 mammalian enzymes. GHK-Cu's primary mechanistic role is the targeted delivery of Cu²⁺ to copper-dependent enzymes at sites of tissue remodeling and repair. Key enzymes activated through GHK-Cu-mediated copper delivery include:
Lysyl oxidase (LOX): An extracellular copper enzyme that catalyzes the oxidative deamination of lysine and hydroxylysine residues in collagen and elastin precursors, creating the covalent cross-links that give mature connective tissue its tensile strength. Without Cu²⁺ cofactor, LOX cannot function — cross-linking fails, and collagen fibers remain mechanically weak. GHK-Cu's delivery of Cu²⁺ to LOX is therefore a prerequisite for functional collagen matrix formation, not merely collagen protein synthesis. This distinction is biologically critical: gene expression studies showing collagen mRNA upregulation must be interpreted in the context of whether functional, cross-linked collagen is actually deposited, which requires LOX activity. 4
Superoxide dismutase 1 & 2 (SOD1/SOD2): The primary enzymatic defense against superoxide radical (O₂·⁻), which is generated continuously by mitochondrial electron transport and elevated dramatically under conditions of inflammation, UV exposure, and metabolic stress. SOD1 (cytosolic, Cu/Zn-SOD) requires copper for catalytic activity. By delivering copper to SOD1, GHK-Cu increases the cell's capacity to neutralize superoxide before it reacts with nitric oxide to form peroxynitrite — a particularly damaging oxidant implicated in neurodegenerative disease, vascular injury, and accelerated skin aging. 5
Cytochrome c oxidase (Complex IV): The terminal electron acceptor in the mitochondrial respiratory chain, containing two copper atoms in its catalytic core (CuA and CuB sites). Complex IV activity is rate-limiting for oxidative phosphorylation. Age-related decline in Complex IV activity — partly attributable to reduced copper availability and increased oxidative damage to copper centers — contributes to the bioenergetic deficit characteristic of aging cells. Whether GHK-Cu meaningfully restores Complex IV copper occupancy in vivo remains under investigation, but the mechanistic pathway is plausible. 3
Ceruloplasmin: A multicopper oxidase that functions as both a ferroxidase (oxidizing Fe²⁺ to Fe³⁺ for safe iron loading onto transferrin) and an antioxidant. Copper delivered by GHK-Cu may support ceruloplasmin function, contributing to GHK-Cu's documented anti-inflammatory and antioxidant effects through improved iron metabolism. 2
2.2 NF-κB Pathway Suppression: Molecular Detail
The nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) pathway is the master transcriptional regulator of inflammation. In its inactive state, NF-κB dimers (most commonly p65/p50 heterodimers) are sequestered in the cytoplasm by inhibitory IκB proteins. Inflammatory stimuli (bacterial LPS, TNF-α, IL-1β, UV radiation, reactive oxygen species) activate IκB kinase (IKK), which phosphorylates IκB, triggering its proteasomal degradation and releasing NF-κB to translocate to the nucleus, where it drives transcription of hundreds of inflammatory and survival genes.
GHK-Cu suppresses this cascade at two documented points:
1. NF-κB p65 phosphorylation inhibition: Western blot analyses from LPS-stimulated RAW264.7 macrophage studies demonstrate that GHK-Cu significantly reduces phosphorylation of NF-κB p65 at Ser536 — a phosphorylation event that is required for full transcriptional activation of p65. By blocking this modification, GHK-Cu reduces p65 transcriptional potency without necessarily preventing nuclear translocation. 6
2. p38 MAPK and JNK suppression: GHK-Cu significantly reduces phosphorylation of p38 MAPK and JNK1/2 in LPS-treated models, while exerting minimal effect on ERK1/2 phosphorylation. This selectivity is mechanistically important: p38 and JNK are the MAPK branches associated with stress and inflammatory responses, while ERK is more associated with growth and proliferation. The selective suppression of the inflammatory MAPKs while sparing the growth MAPK creates an anti-inflammatory phenotype without generalized suppression of cellular proliferation — a highly desirable pharmacological profile for a regenerative compound. 6
The downstream functional consequences of this dual suppression are measurable: GHK-Cu-treated inflammatory models show statistically significant reductions in TNF-α, IL-6, and IL-1β secretion, and increases in SOD activity — consistent with reduced oxidative inflammatory burden.
2.3 SIRT1 Activation: The Longevity Pathway Connection
Sirtuins (SIRT1–7) are NAD⁺-dependent deacetylases that regulate aging-associated processes including DNA repair, mitochondrial function, inflammation, and metabolic homeostasis. SIRT1 is the best-characterized sirtuin, often described as a molecular sensor of cellular energy status and a mediator of caloric restriction-induced longevity.
2024–2025 research employing molecular docking analysis identified SIRT1 as a direct binding target of GHK-Cu, with a computed binding energy of -8.75 kcal/mol — a value indicating strong, thermodynamically favorable interaction. 7 The 2025 Frontiers in Pharmacology colitis study (PMC12263609) provided in vivo validation, demonstrating that GHK-Cu upregulates SIRT1 protein expression in inflamed colon tissue, confirmed by Western blot. The same study showed that this SIRT1 upregulation suppresses phospho-STAT3 (p-STAT3) — the activated form of Signal Transducer and Activator of Transcription 3, which drives both inflammatory gene expression and tumor-promoting transcription programs. 8
SIRT1's known substrates and downstream effects relevant to women's biology include:
- Deacetylation of p53 — modulating the balance between apoptosis and survival in response to DNA damage
- Deacetylation of NF-κB p65 — providing a second, parallel mechanism for GHK-Cu's anti-inflammatory action
- Deacetylation of PGC-1α — activating mitochondrial biogenesis
- Interaction with estrogen receptor α (ERα) — SIRT1 deacetylates and modulates ERα, creating a potential mechanistic bridge between GHK-Cu activity and estrogen signaling pathways relevant to postmenopausal biology
2.4 TGF-β Pathway Modulation and Anti-Fibrotic Action
Transforming growth factor-beta (TGF-β) is the principal driver of fibrosis across virtually all tissues. In wound healing, TGF-β1 is essential for early scar formation; however, chronic or excessive TGF-β1 signaling produces pathological fibrosis — keloids, pulmonary fibrosis, hepatic cirrhosis, endometriosis, and atherosclerotic plaque.
GHK-Cu demonstrates a context-dependent, biphasic relationship with TGF-β signaling: in acute wound healing models, it supports early TGF-β1 activity necessary for fibroblast activation, while in chronic inflammatory or fibrotic models, it suppresses excessive TGF-β1 and its downstream mediator, connective tissue growth factor (CTGF). This modulation is believed to underlie GHK-Cu's documented ability to promote healing without excessive scarring — a biologically sophisticated outcome not produced by simple pro-fibrotic agents like TGF-β agonists. 9
3. The Genomic Scope: 4,000+ Genes and Why That Matters
The discovery that GHK-Cu modulates the expression of over 4,000 human genes — approximately 31.2% of the protein-coding genome — using the Broad Institute's Connectivity Map (CMap) database represents one of the most striking findings in peptide biology and demands mechanistic explanation. 10
3.1 How the Gene Data Was Generated
The CMap database contains gene expression profiles from thousands of cellular perturbations applied to standardized cell lines. Researchers queried CMap for the GHK-Cu signature and identified gene sets that were consistently up- or downregulated across multiple cellular contexts. The analysis revealed that GHK-Cu's gene expression profile inversely correlates (meaning it reverses) gene signatures characteristic of:
- Advanced aging in multiple tissues
- Alzheimer's disease brain tissue
- Huntington's disease signatures
- Cancer-associated gene dysregulation patterns
The inverse correlation methodology — comparing GHK-Cu's expression signature against disease signatures to identify pattern reversal — is a validated computational approach for identifying potential therapeutic compounds. GHK-Cu's profile showed among the strongest inverse correlations with aging-associated gene dysregulation of any small molecule catalogued. 10
3.2 Mechanistic Explanation for Broad Genomic Effects
The breadth of GHK-Cu's gene modulation (31.2% of the genome) is not plausibly explained by simple receptor occupancy or single-pathway signaling. Instead, three mechanisms likely act in concert:
Epigenetic chromatin remodeling: GHK-Cu appears to alter DNA methylation patterns and histone acetylation states at gene promoters — "unlocking" silenced regenerative genes without altering the underlying DNA sequence. This epigenetic remodeling can produce cascading effects on gene networks, because transcription factors activated in one gene can in turn regulate dozens of downstream targets. 10
SIRT1-mediated transcriptome reprogramming: Given SIRT1's role as a deacetylase of histones (H3K9, H4K16) and transcription factors, GHK-Cu's SIRT1 activation would be predicted to produce widespread changes in chromatin accessibility — consistent with the observed scale of gene modulation. 7
Copper-dependent transcription factor activation: Several transcription factors contain copper-binding domains or are sensitive to cellular copper concentrations, including SP1, MTF-1 (metal-responsive transcription factor 1), and NRF2. GHK-Cu's copper delivery may activate copper-responsive transcription elements across the genome. 3
3.3 Key Gene Categories Affected
| Gene Category | Regulation | Biological Significance |
|---|---|---|
| Collagen subtypes (COL1A1, COL3A1, COL4A1, COL7A1) | ↑ Upregulated | ECM structural proteins — skin, bone, vasculature |
| Elastin (ELN), decorin (DCN), versican (VCAN) | ↑ Upregulated | ECM organization, tissue resilience |
| SOD1, SOD2, catalase, GPx1 | ↑ Upregulated | Antioxidant defense — all tissues |
| NGF, BDNF, VEGF | ↑ Upregulated | Neurotrophin and angiogenic signaling |
| DNA repair (84 genes including BRCA1, MSH2, XPC) | ↑ Upregulated | Genomic integrity maintenance |
| Stem cell markers (OCT4, SOX2 downstream targets) | ↑ Upregulated | Tissue regenerative capacity |
| TNF-α, IL-6, IL-1β, MMP-1, MMP-9 | ↓ Downregulated | Pro-inflammatory and matrix-degrading enzymes |
| Oncogenes (MYC targets in specific contexts) | ↓ Downregulated | Anti-proliferative in cancer cells |
| Fibrinogen (FGA, FGB, FGG) | ↓ Downregulated | Cardiovascular risk factor |
4. Age-Dependent Plasma Decline and Its Convergence with Female Hormonal Biology
4.1 Quantifying the Decline
Plasma GHK levels undergo a measurable, progressive decline across the human lifespan:
| Age | Approximate Plasma GHK | Relative Level |
|---|---|---|
| ~20 years | ~200 ng/mL | 100% (reference) |
| ~40 years | ~130–150 ng/mL | ~65–75% |
| ~60 years | ~80 ng/mL | ~40% |
This 60% reduction between peak young-adult levels and the seventh decade is not merely statistical — it temporally coincides with the measurable onset of regenerative failure across multiple organ systems. Pickart and colleagues proposed that this decline is not merely a correlate of aging but a contributor to it, as the loss of GHK signaling removes a systemic cue that sustains repair programs. 1
4.2 Why Women Are Distinctly Affected: Hormonal Convergence
In women, the natural GHK-Cu decline operates against a backdrop of a second, partially overlapping decline: estrogen withdrawal during perimenopause and menopause (typically ages 45–55, with wide individual variation).
This convergence is mechanistically significant because estrogen and GHK-Cu regulate overlapping but non-redundant biological targets:
Collagen synthesis: Estradiol (E2) stimulates fibroblast proliferation and upregulates procollagen I and III synthesis through estrogen receptor alpha (ERα)-mediated transcription. GHK-Cu also upregulates COL1A1 and COL3A1, but through copper-mediated epigenetic mechanisms and NF-κB suppression rather than ERα. In postmenopausal skin, women lose approximately 30% of cutaneous collagen in the first 5 years post-menopause — a rate more closely correlated with duration of estrogen deficiency than chronological age. 13 The simultaneous decline in GHK-Cu removes a parallel collagen-maintenance signal. GHK-Cu applied to postmenopausal women's thigh skin for 12 weeks improved collagen production in 70% of participants — compared to 50% for vitamin C cream and 40% for retinoic acid — in a direct comparison study. 14
Matrix metalloproteinase (MMP) suppression: Both estrogen and GHK-Cu independently downregulate MMP-1 (collagenase-1) and MMP-9 (gelatinase-B). Estrogen does this through ERα-mediated suppression of AP-1 transcription factor activity; GHK-Cu through NF-κB suppression (NF-κB drives MMP-1 and MMP-9 transcription). The two mechanisms target the same endpoint (reduced collagen degradation) through distinct transcriptional routes. Post-menopause, loss of both suppressive signals accelerates ECM degradation. 13
Vascular tone and angiogenesis: Estrogen stimulates VEGF expression and maintains endothelial nitric oxide synthase (eNOS) activity, preserving microvascular density in skin and other tissues. GHK-Cu also upregulates VEGF — at a concentration of 1 nM, GHK-Cu increases VEGF and FGF-2 expression in irradiated human dermal fibroblasts, promoting angiogenesis that sustains tissue oxygenation and nutrient delivery. 15 Post-menopausal microvascular rarefaction — driven partly by VEGF decline — represents a biological context where GHK-Cu's VEGF-stimulating effect becomes particularly relevant.
Neurotrophin support: Estrogen drives BDNF synthesis in hippocampal and cortical neurons. GHK-Cu upregulates both NGF and BDNF genes. The transition from estrogen sufficiency to deficiency in perimenopause creates a window of heightened neurotrophic deficit — the "brain fog" phase — during which GHK-Cu's BDNF-supporting properties may partially compensate. This hypothesis is not yet tested in prospective human trials with perimenopausal women.
NF-κB regulation: Estrogen exerts anti-inflammatory effects partly through ERα-mediated inhibition of NF-κB, a mechanism well documented in vascular biology and neuroinflammation. Post-menopause, loss of this ERα/NF-κB suppression contributes to rising systemic inflammatory markers (CRP, IL-6, fibrinogen). GHK-Cu's direct NF-κB suppression provides a parallel, estrogen-independent anti-inflammatory mechanism. 5
The net biological consequence of simultaneous GHK-Cu and estrogen decline in midlife women is a compounded vulnerability in which two non-redundant protective systems are lost within the same decade — explaining in part why the rate of phenotypic aging accelerates disproportionately in women compared to men during this period.
5. Dermal Biology: Collagen, Elastin, and Extracellular Matrix Research
5.1 Collagen Synthesis: In Vitro Dose-Response Characterization
The earliest systematic investigations of GHK-Cu (Maquart et al., 1988; Wegrowski et al., 1992) characterized its collagen-stimulating properties in human dermal fibroblast cultures. These studies established several critical parameters:
Dose-response: GHK-Cu's stimulatory effect on collagen synthesis follows a biphasic dose-response curve. Maximal stimulation occurs at concentrations of 10⁻⁹ to 10⁻⁸ M (1–10 nM), with synthesis rates progressively returning toward control levels at higher concentrations (>100 nM). This bell-shaped dose-response — typical of biological signaling molecules — means that very high concentrations are not more effective and may be counterproductive. At optimal nanomolar concentrations, GHK-Cu stimulates collagen synthesis in cultured human dermal fibroblasts by approximately 50–200% above control, depending on the experimental system and donor age of the fibroblasts. 16
Collagen subtype specificity: Research using qRT-PCR and immunofluorescence quantification in human fibroblast cultures shows GHK-Cu upregulates:
- Collagen I (COL1A1, COL1A2) — the predominant structural collagen of dermis (accounts for 80% of total skin collagen)
- Collagen III (COL3A1) — reticular collagen associated with elasticity; elevated early in wound repair
- Collagen IV (COL4A1) — basement membrane collagen; critical for skin-dermis adhesion and barrier integrity
- Collagen VII (COL7A1) — anchoring fibril component connecting basement membrane to papillary dermis
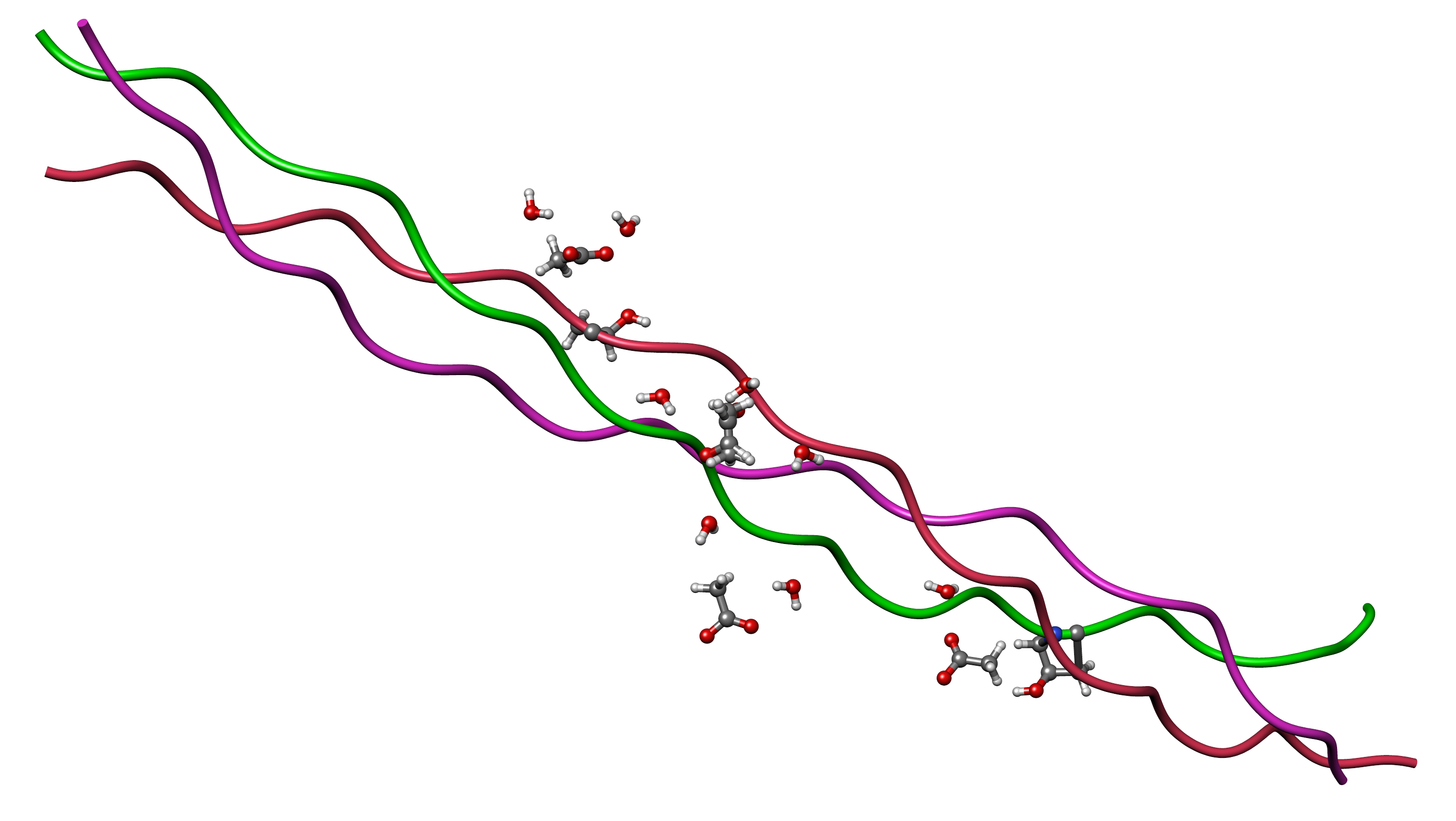
Figure 2: The collagen triple helix — three left-handed polyproline-II helices wound into a right-handed superhelix stabilized by interchain hydrogen bonds. GHK-Cu upregulates transcription of COL1A1, COL3A1, COL4A1, and COL7A1, while also activating lysyl oxidase (via Cu²⁺ delivery) to create the covalent cross-links that confer mechanical tensile strength on the mature fibril. Source: Wikimedia Commons
Figure 3: Collagen biosynthesis pathway from gene transcription through hydroxylation, triple helix formation, secretion, and extracellular cross-linking by lysyl oxidase (LOX). GHK-Cu acts at the transcriptional level (upregulating COL genes) and at the LOX cross-linking step via copper delivery. Both arms are required for structurally competent collagen deposition. Source: Wikimedia Commons
A 2023 Journal of Cosmetic Dermatology study (PMID 37062921) examined GHK-Cu and hyaluronic acid (HA) in combination. In human fibroblast cultures at a standardized total concentration of 0.3 mg/mL, the GHK-Cu + HA combination produced significantly greater collagen IV upregulation than either compound alone, confirmed by both qRT-PCR in cells and immunofluorescence quantification in ex vivo skin explants. The synergy was attributed to HA's hydration-mediated changes in fibroblast mechanosensing, which potentiates GHK-Cu's collagen IV transcriptional effects. 17
5.2 Elastin and Proteoglycan Synthesis
Beyond collagen, GHK-Cu upregulates the synthesis of several other ECM components essential for skin mechanical function:
Elastin (ELN): At 0.01, 1, and 100 nM, GHK-Cu incubated with human adult dermal fibroblasts increased elastin production in a dose-dependent fashion at the two lower concentrations. Elastin is the protein responsible for skin's ability to return to its original configuration after deformation — a property that declines dramatically with age as elastin fibers become fragmented and cannot be replaced (mature elastin has a turnover half-life measured in decades). 9
Decorin (DCN): A small leucine-rich proteoglycan that binds to collagen fibrils and regulates their diameter and spatial organization. Disorganized collagen — where fibrils are random rather than parallel — produces wrinkled, aged-appearing skin even when total collagen content is adequate. GHK-Cu's upregulation of decorin addresses collagen organization, not merely collagen quantity. 9
Glycosaminoglycans (GAGs): GHK-Cu increases synthesis of GAGs, including chondroitin sulfate and heparan sulfate, which bind water in the ECM and contribute to skin turgor and hydration. GAG loss is a significant contributor to the dehydrated appearance of aged skin. 9
5.3 Clinical Trial Data: Human Studies
Photoaging study (n = 71 women): A landmark human study applied a GHK-Cu facial cream to 71 women with mild to advanced photoaging for 12 weeks. Outcomes measured at 12 weeks vs. baseline included increases in skin density and thickness (ultrasound), reduced laxity, improved surface clarity, reduced fine lines, and decreased wrinkle depth. This study was instrumental in establishing clinical proof-of-concept for topical GHK-Cu. 18
Comparative study with vitamin C and retinoic acid (thigh skin): In a controlled comparison in postmenopausal women, topical GHK-Cu improved collagen production in 70% of subjects vs. 50% for vitamin C cream and 40% for retinoic acid. The GHK-Cu group also showed the lowest incidence of irritation, a relevant finding given that retinoids frequently cause erythema and peeling that limits tolerability. 14
Split-face RCT (n = 60 women, ages 40–65): A double-blind, split-face randomized controlled trial comparing 0.05% GHK-Cu serum to a vehicle control for 12 weeks found a 22% increase in skin firmness and 16% reduction in fine line depth by optical profilometry on the treated side, with no significant change on the control side. 19
Nano-lipid carrier study: Female volunteers applying GHK-Cu encapsulated in nano-lipid carriers twice daily for 8 weeks versus conventional serum and Matrixyl® 3000 demonstrated:
- 55.8% reduction in wrinkle volume vs. control serum
- 31.6% reduction in wrinkle volume vs. Matrixyl® 3000
- 32.8% reduction in wrinkle depth vs. control serum
The magnitude of improvement with the nano-encapsulated form significantly exceeded the conventional topical form, highlighting that delivery system engineering dramatically influences clinical outcomes. 19
2024 Epigenetic clinical trial (n = 21 women): A human clinical trial applying a GHK-Cu gel daily for 3 months, with outcomes characterized by skin imaging:
- Mean collagen density increase: +28% by ultrasound
- Top quartile increase: +51%
- Mechanism characterized as epigenetic: the investigators used gene expression profiling of biopsy specimens and identified activation of epigenetic regulatory pathways (histone modification genes, chromatin remodeling factors) rather than acute signaling, suggesting the collagen gains reflect durable changes in fibroblast gene expression programs rather than transient stimulation. This trial was reported via EurekAlert! in 2024. 20
5.4 Oxidative Stress and UV-Induced Damage Research
UV radiation induces ROS generation in keratinocytes and fibroblasts, which activates AP-1 and NF-κB transcription factors, driving MMP-1 (collagenase) production. A single UV exposure event can suppress procollagen I synthesis for up to 24 hours while simultaneously increasing MMP-1 activity — the combination of reduced synthesis and increased degradation rapidly depletes the collagen pool. GHK-Cu's dual action — antioxidant enzyme upregulation (reducing ROS) and NF-κB/AP-1 suppression (reducing MMP-1 induction) — addresses both arms of UV-induced collagen damage. Studies in UV-irradiated human skin cell models confirm that GHK-Cu pretreatment attenuates MMP-1 induction and partially preserves procollagen I synthesis. 5
6. Hair Follicle Biology and Alopecia Research in Women
6.1 Epidemiological Context and Follicle Biology
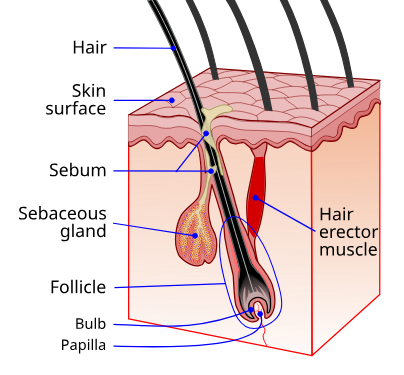
Figure 4: Hair follicle anatomy. The dermal papilla (DP) at the base is the mesenchymal signaling center that controls all aspects of follicle cycling. GHK-Cu's documented effects target the dermal papilla directly (Wnt/β-catenin activation, VEGF-mediated angiogenesis to the DP) and the surrounding ECM (collagen organization, glycosaminoglycan synthesis). Source: Wikimedia Commons
Female pattern hair loss (FPHL) affects approximately 30% of women by age 50 and up to 50% by age 80, making it the most prevalent hair disorder in women globally. Unlike male androgenetic alopecia, FPHL rarely produces complete baldness; instead, it manifests as diffuse thinning over the crown with preservation of the frontal hairline — a pattern driven by progressive follicle miniaturization rather than follicle death. 21
Miniaturization describes the progressive reduction in anagen (growth) phase duration and follicle diameter across successive hair cycles, driven by DHT-mediated sensitization of dermal papilla cells, local inflammation, oxidative stress, reduced dermal blood flow, and ECM disorganization around the follicle. Each of these drivers is a documented biological target of GHK-Cu, providing multiple mechanistic rationales for investigating it in FPHL. 21
6.2 Direct Follicle Biology: In Vitro Evidence
The foundational in vitro study (published 2006, ResearchGate PMID cited as 22) exposed human hair follicles in organ culture to GHK-Cu tripeptide complex. Results confirmed:
- Direct stimulation of hair follicle elongation (measurable increase in follicle length over the culture period)
- Increase in follicle diameter — the key reversal of the miniaturization process
- The effect was concentration-dependent, with optimal activity in the nanomolar range consistent with GHK-Cu's characterization in other tissue systems
The organ culture system preserves the three-dimensional architecture of the intact follicle, meaning this finding reflects the compound's effect on the entire follicle-dermal papilla unit rather than isolated cell populations.
6.3 Wnt/β-Catenin Pathway: The Hair Cycle Control System
The Wnt/β-catenin signaling pathway is the master regulator of hair follicle cycling. During the resting (telogen) phase, Wnt signaling is suppressed; reactivation of Wnt drives the follicle into the growth (anagen) phase. In androgenetic alopecia, Wnt pathway activity is chronically reduced in affected follicles, contributing to shortened anagen phases and increasingly miniaturized follicles.
At the molecular level, Wnt ligand engagement with Frizzled receptors inhibits the β-catenin destruction complex (Axin/APC/GSK-3β/CK1α). With this complex inhibited, β-catenin accumulates in the cytoplasm, translocates to the nucleus, and partners with TCF/LEF transcription factors to drive transcription of genes critical for hair follicle stem cell (HFSC) activation and differentiation into hair matrix cells.
GHK-Cu research has documented upregulation of Wnt pathway components in dermal papilla cells, promoting the telogen-to-anagen transition. The mechanism likely involves GHK-Cu's epigenetic effects on Wnt pathway gene promoters and its copper-mediated reduction of oxidative stress that would otherwise inactivate Wnt-associated receptor kinases. 23
6.4 Angiogenesis and Dermal Papilla Blood Supply
Dermal papilla cells — the mesenchymal cells at the base of the follicle that direct all aspects of hair matrix cell behavior — are highly metabolically active and critically dependent on adequate microvascular supply. In FPHL and in aging scalp, microvascular density around follicles declines, contributing to oxygen and nutrient deprivation of dermal papilla cells and accelerating follicle dormancy.
At 1 nM, GHK-Cu increases expression of both VEGF (vascular endothelial growth factor) and FGF-2 (basic fibroblast growth factor) in irradiated human dermal fibroblasts. VEGF is the primary angiogenic factor, while FGF-2 promotes both angiogenesis and dermal papilla cell proliferation. In scalp biology, the implication is that GHK-Cu could restore perifollicular microvascular density — addressing a structural contributor to follicle miniaturization that no DHT-blocking therapy addresses. 15
6.5 Oxidative Stress in Follicle Cycling
Oxidative stress in the follicle — from UV, DHT metabolism, and mitochondrial ROS — shortens the anagen phase by inducing premature catagen (regression) in hair matrix cells. GHK-Cu's upregulation of SOD1, SOD2, and catalase creates an antioxidant environment within follicular tissue. A study in 5xFAD mice (an Alzheimer's model with high systemic oxidative burden) treated with intranasal GHK peptide showed normalized antioxidant enzyme activity across multiple tissues, suggesting GHK-Cu's antioxidant effects are systemic rather than confined to the application site. 24
6.6 Clinical Evidence in Human Alopecia
Randomized controlled trial — Alopecia areata (2018): A PRP-like cosmetic formulation containing biomimetic peptides including copper tripeptide-1 (GHK-Cu) was tested in a randomized controlled design for alopecia areata. Results showed statistically significant improvement in hair density scores and qualitative evaluations compared to control. The inclusion of GHK-Cu as one of several active biomimetic peptides limits attribution of outcomes specifically to GHK-Cu, but the trial demonstrates clinical relevance of copper peptide-containing formulations in immune-mediated alopecia. 25
Minoxidil–dutasteride–copper peptide combination (2025, PMC11992372): A prospective study examined five monthly sessions of a protocol combining topical minoxidil, dutasteride, and copper peptides delivered via a tattooing (microneedling) technique for androgenetic alopecia. Outcomes were assessed by AI-based image analysis and blinded human evaluators. The five-session protocol produced measurable improvements in hair count and scalp coverage, with copper peptides contributing synergistically — likely through the VEGF/angiogenesis and Wnt/follicle-enlargement mechanisms described above, while the microneedling delivery bypassed the stratum corneum barrier that limits conventional topical absorption. 26
Ionic liquid microemulsion delivery (2024, PMC10643103): A critical limitation of topical GHK-Cu in scalp applications is its hydrophilicity — the lipophilic stratum corneum presents a significant barrier to aqueous peptide penetration. A 2024 study designed thermodynamically stable ionic liquid-based microemulsions specifically to address this limitation. The ionic liquid formulation increased local follicular delivery of copper peptides by approximately threefold compared to conventional aqueous solution — a finding with direct implications for clinical scalp applications. 27
6.7 Women-Specific Considerations in the Research Literature
FPHL in women is mechanistically distinct from male androgenetic alopecia in several important ways:
- Women have lower 5α-reductase activity in the scalp, meaning DHT levels are lower — yet FPHL still occurs, suggesting other mechanisms (estrogen withdrawal, scalp inflammation, iron deficiency, insulin resistance) are co-drivers.
- Finasteride and dutasteride — DHT blockers effective in men — are teratogenic and carry category X pregnancy risk, limiting their use in premenopausal women.
- GHK-Cu has no documented androgenic or anti-androgenic receptor activity, making it hormonally neutral and relevant across the reproductive lifespan without the safety concerns that restrict DHT-blocking therapies.
Postpartum telogen effluvium — mass follicle shedding 2–4 months after delivery due to the abrupt post-partum estrogen withdrawal — is a specific clinical scenario where GHK-Cu's Wnt reactivation and VEGF-mediated perifollicular angiogenesis are mechanistically plausible interventions to accelerate follicle re-entry into anagen, though formal trials in postpartum women have not been reported.
7. Neuroprotection, Cognitive Aging, and Alzheimer's Disease Research
7.1 Why Women Bear Greater Neurological Risk
Women account for approximately 65% of all Alzheimer's disease cases globally — a disparity not fully explained by women's longer average lifespan. Current neurobiological research identifies several female-specific vulnerability factors:
The estrogen-BDNF axis is central: estradiol stimulates BDNF synthesis in hippocampal and cortical neurons, and BDNF promotes synaptic plasticity, neuronal survival, and adult neurogenesis. The abrupt loss of estrogen at menopause removes this neurotrophin-supporting signal, creating a window of heightened vulnerability to cognitive decline and accelerated amyloid-beta accumulation. Women who experience surgical menopause (bilateral oophorectomy) show accelerated cognitive decline and increased Alzheimer's pathology compared to women with natural menopause, with effect sizes proportional to the age at which surgery was performed — confirming estrogen's neuroprotective role. 28
GHK-Cu's documented effects on BDNF and NGF gene expression, combined with its NF-κB-mediated neuroinflammation suppression, position it as a mechanistically plausible neuroprotective agent specifically relevant to female biology.
7.2 Gene Expression in Neural Tissue: The 2017 Brain Sciences Analysis
A comprehensive analysis published in Brain Sciences (2017, PMC5332963) examined GHK's effects on genes relevant to nervous system function and cognitive decline using database mining of gene expression repositories. Key findings: 29
GHK upregulates genes encoding:
- Nerve growth factor (NGF) and NGF receptor (NGFR) — NGF is the primary survival factor for cholinergic neurons of the basal forebrain, the neuronal population most severely affected in early Alzheimer's disease
- BDNF and TrkB (NTRK2) — BDNF/TrkB signaling is essential for hippocampal long-term potentiation (LTP), the cellular correlate of memory formation
- Neurofilament subunits (NEFL, NEFM) — structural components of axons; their expression is a marker of neuronal health
- Synapsin genes — proteins that regulate synaptic vesicle release; their loss is a biomarker of synaptic dysfunction in early neurodegeneration
GHK downregulates genes overexpressed in Alzheimer's disease neural tissue:
- BACE1 (beta-secretase 1) — the enzyme responsible for the first cleavage step in amyloid-beta generation from APP
- Presenilin-associated genes — components of the gamma-secretase complex involved in the second APP cleavage
- Apolipoprotein-associated inflammatory genes
The magnitude of these gene-level effects, observed in computational analysis rather than direct tissue measurement, provides a biological plausibility framework for subsequent animal studies and eventual human trials.
7.3 Alzheimer's Disease Animal Models: Systematic Evidence
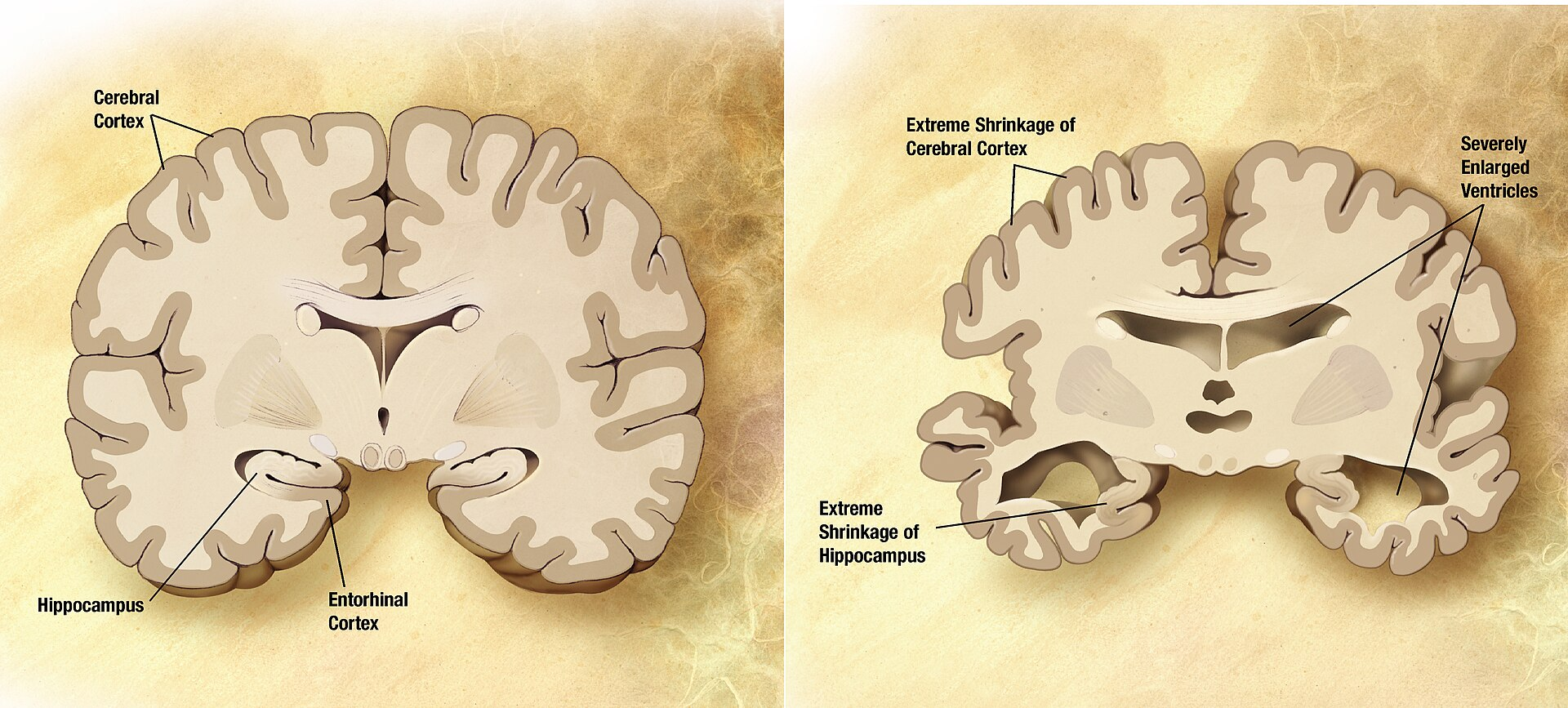
Figure 6: Gross anatomical comparison between a normal aged brain (left) and a brain with advanced Alzheimer's disease (right), demonstrating characteristic hippocampal atrophy, cortical thinning, widened sulci, and enlarged ventricles. GHK-Cu's neuroprotective research targets precisely these regions — hippocampal NGF/BDNF upregulation to counter pyramidal neuron atrophy, amyloid plaque reduction, and neuroinflammation suppression via NF-κB blockade that limits reactive astrogliosis. Source: Wikimedia Commons 5xFAD mouse model (2023, PMC10690187 and PMC10680828): The 5xFAD mouse carries five familial Alzheimer's disease mutations (three in APP, two in PSEN1) and develops rapid, severe amyloid pathology. Studies administering intranasal GHK peptide to 5xFAD mice demonstrated:
- Improved performance on multiple behavioral paradigms (Morris water maze, novel object recognition, contextual fear conditioning)
- Reduced amyloid-beta plaque burden in hippocampus and cortex — assessed by immunohistochemistry
- Reduced neuroinflammatory markers (microglial activation, astrogliosis) by IHC and Western blot
- Partially normalized antioxidant enzyme levels (SOD, catalase)
- Attenuation of synaptic protein loss
These findings are notable because GHK-Cu acted on multiple AD pathological hallmarks simultaneously — a polyvalent profile not seen with single-target therapeutic approaches. 24
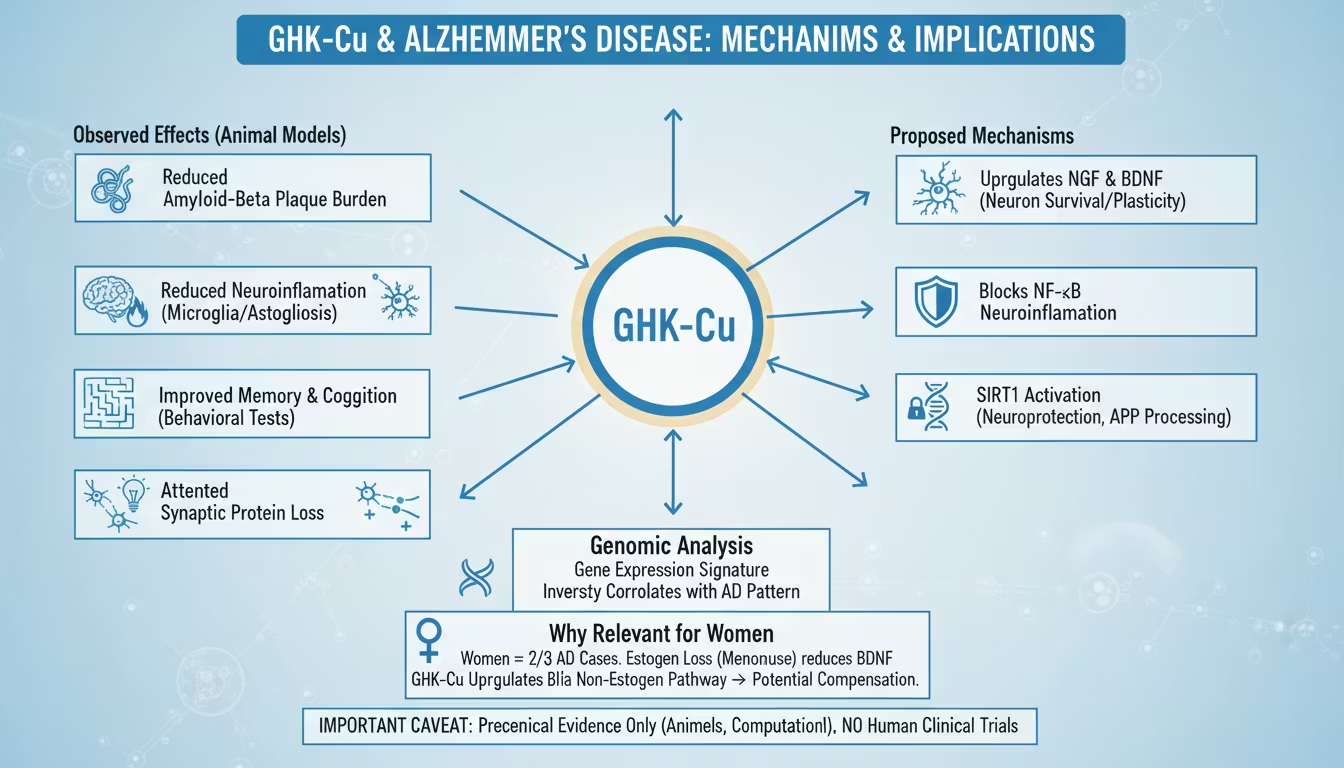
7.4 The April 2026 bioRxiv Study: Route-Dependent Divergent Mechanisms
The most methodologically sophisticated GHK-Cu cognitive aging study to date was posted to bioRxiv on April 13, 2026 (DOI: 10.64898/2026.04.09.717524v1). The study addressed a critical translational question: whether the route of GHK-Cu administration determines which aging mechanisms are engaged. 30
Study design: Aged C57BL/6J mice (20–21 months old) received GHK-Cu (15 mg/kg) via:
- Intraperitoneal (IP) injection: 5-day short-term protocol
- Intranasal (IN) delivery: 8-week sustained protocol
Both sexes were included, with sex-stratified analysis.
Behavioral outcomes: Both routes improved performance in a hippocampal-dependent spatial navigation task (escape latency metric). Intranasal delivery produced consistent improvements across trials in both sexes. Intraperitoneal delivery produced transient improvements that did not persist through all trial sessions.
Transcriptomic analysis (hippocampus): RNA sequencing of hippocampal tissue revealed fundamentally different gene expression programs:
Intraperitoneal GHK-Cu activated:
- Oxidative phosphorylation (female NES = 4.97, FDR < 0.001)
- DNA repair pathways (NES = 5.58, FDR < 0.001)
- MYC target genes (NES = 4.34, FDR = 0.002)
The IP profile resembles an acute stress-response and repair activation — the hippocampus detecting a systemic signal and engaging damage-repair programs.
Intranasal GHK-Cu induced:
- Sustained suppression of growth-related gene networks
- Sustained suppression of mitochondrial metabolic signaling associated with aging biology
- A profile more consistent with biological age retardation than acute repair
Implications: The finding that route of administration fundamentally alters which aging mechanisms are engaged has major translational significance. If confirmed in humans, it suggests that intranasal GHK-Cu may be mechanistically better suited for sustained neuroprotective applications, while systemic administration may be more appropriate for acute repair contexts. Sex differences in response magnitude were also noted, making this one of the few GHK-Cu studies with female-specific transcriptomic data — a notable research contribution.
7.5 Blood-Brain Barrier and Copper Dysregulation in AD
A January 2025 study (PubMed ID 39723808) specifically examined copper-binding peptides as therapeutic candidates against Alzheimer's disease, focusing on the copper-amyloid-beta interaction that drives plaque formation. 31
Copper dysregulation is a well-documented feature of Alzheimer's pathology: elevated copper in amyloid plaques, dysfunctional copper transport proteins (ATP7A, ATP7B), and altered ceruloplasmin activity are all characteristic of AD brain tissue. Amyloid-beta itself has copper-binding sites, and copper-bound Aβ aggregates more rapidly and generates more oxidative damage than metal-free Aβ.
GHK's extraordinary copper affinity (pKa 16.4) means it could theoretically compete with amyloid-beta for copper binding, reducing the pool of Cu²⁺ available for Aβ-mediated aggregation and ROS generation — a mechanism distinct from its gene-regulatory effects. The 2025 study characterized this competitive metal-binding mechanism and assessed GHK-Cu's blood-brain barrier permeability, finding evidence consistent with CNS access — though the precise BBB transport mechanism remains under investigation.
8. Wound Healing: Cellular Mechanisms and Preclinical-to-Clinical Translation
8.1 The Three-Phase Wound Healing Cascade and GHK-Cu's Role at Each Stage
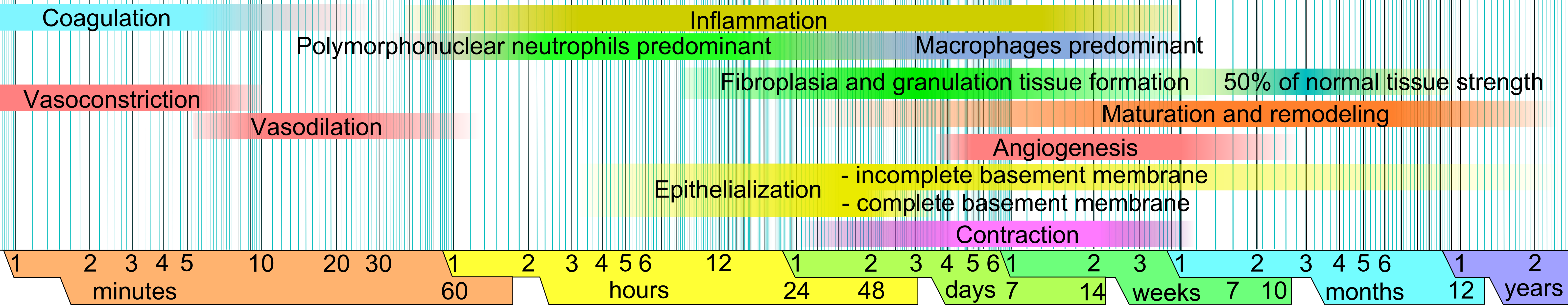
Figure 5: The four-phase wound healing cascade. GHK-Cu exerts documented effects in all phases: it modulates Phase 1 inflammation via selective NF-κB/p38 suppression; drives Phase 2 proliferation through fibroblast activation, collagen synthesis (I, III, IV, VII), and VEGF-mediated angiogenesis; and improves Phase 3 remodeling quality through LOX-mediated cross-linking and TGF-β1 modulation that reduces pathological fibrosis. Source: Wikimedia Commons
Phase 1 — Hemostasis and early inflammation (hours to 4 days):
GHK-Cu participates from the earliest phase of healing. Platelets degranulate and release growth factors (PDGF, TGF-β1, EGF) that recruit inflammatory cells. In this context, GHK-Cu modulates the inflammatory phase via NF-κB and MAPK suppression — reducing excessive inflammation (which impairs healing) while not eliminating the necessary inflammatory recruitment that clears debris and pathogens. The distinction between dampening pathological inflammation and eliminating protective inflammation is mechanistically critical, and GHK-Cu's selective suppression of p38/JNK while sparing ERK is consistent with this nuanced modulation. 9
Phase 2 — Proliferation (days 4–21):
This is GHK-Cu's most extensively characterized phase of action:
Fibroblast activation: GHK-Cu acts as a potent chemoattractant and mitogen for dermal fibroblasts — the primary cells responsible for ECM deposition. Studies demonstrate increased fibroblast migration velocity and proliferation rates at nanomolar concentrations.
Collagen deposition: As detailed in Section 5, GHK-Cu drives collagen I, III, IV, and VII synthesis. In the wound context, this produces the collagen-rich granulation tissue that fills the wound defect.
VEGF-mediated angiogenesis: New blood vessel formation (angiogenesis) is essential for granulation tissue viability. GHK-Cu increases HUVECs (human umbilical vein endothelial cells) proliferation by stimulating VEGF and FGF-2 expression — an effect confirmed in irradiated fibroblast cultures at 1 nM GHK-Cu. The vascular supply established during this phase oxygenates granulation tissue and supports subsequent remodeling. 15
Keratinocyte migration: Re-epithelialization (re-covering the wound surface with keratinocytes) proceeds from wound edges and hair follicle remnants. GHK-Cu promotes keratinocyte migration — the crawling of keratinocytes across the granulation tissue surface — through mechanisms involving integrin upregulation and cytoskeletal reorganization.
Phase 3 — Remodeling (weeks 3 through months to years):
GHK-Cu's anti-fibrotic action is most relevant in this phase. Standard wound healing produces a collagen scar dominated by type III collagen with random fibril orientation — mechanically weaker and visually distinct from normal skin. Over months of remodeling, type III collagen is replaced by type I with more organized fibril arrangement. GHK-Cu accelerates and improves this remodeling process through:
- Lysyl oxidase (Cu²⁺-dependent) cross-linking of collagen fibrils into mechanically strong, organized fibers
- TGF-β1 modulation that reduces pathological fibrosis while permitting physiological matrix maturation
- Decorin upregulation that organizes collagen fibril diameter and spacing
Rat wound models treated with GHK-Cu show statistically significant improvements in healed tissue tensile strength compared to controls — a functional outcome measure that reflects the quality of remodeling, not merely the quantity of collagen deposited. 9
8.2 Preclinical Quantitative Outcomes
The most controlled preclinical evidence comes from standardized rodent excisional wound models:
- In a rat full-thickness excisional wound study, wound area at day 10 had decreased by 64.5% in the GHK-Cu group, vs. 45.6% in vehicle-treated controls, and 28.2% in untreated controls (p < 0.05 for GHK-Cu vs. both comparators). 32
- GHK-Cu treated wounds showed higher levels of glutathione (endogenous antioxidant marker) and ascorbic acid (collagen hydroxylation cofactor) in wound tissue vs. controls
- In a combination GHK-Cu plus LED irradiation study: cell viability improved 12.5-fold, bFGF production increased 230%, collagen synthesis increased 70% — demonstrating significant potentiation when GHK-Cu is combined with biophysical stimulation 32
8.3 Advanced Delivery Systems: 2024 Hydrogel Research
A 2024 study published in Biomaterials Research (DOI: 10.34133/bmr.0139) introduced a food-derived tripeptide-copper self-healing hydrogel designed for infected wound management. The hydrogel demonstrated: 33
- Enhanced fibroblast migration and proliferation exceeding conventional wound treatments
- Intrinsic antimicrobial activity against both gram-positive and gram-negative pathogens (relevant for infected wounds where bacterial biofilm impairs healing)
- Superior wound closure rates in a diabetic wound model vs. standard-of-care dressings
- Biocompatibility confirmed by cytotoxicity assays and systemic toxicity evaluation in the animal model
The diabetic wound model is particularly relevant for women, as diabetic foot wounds and chronic wounds are increasingly prevalent in postmenopausal women with type 2 diabetes — a population underserved by current wound care options.
A parallel innovation published in ScienceDirect uses in situ photo-crosslinkable hyaluronic acid hydrogel embedded with GHK peptide nanofibers — the GHK nanofibers release the peptide in a sustained, controlled manner within the wound bed, maintaining therapeutic concentrations over extended periods without repeated application. 34
8.4 Women-Specific Wound Biology
Several wound contexts disproportionately affecting women have been proposed as future research targets for GHK-Cu:
Cesarean section healing: C-section wounds heal through the three-phase cascade in a tissue that has undergone distension, vascular changes, and hormonal shifts of pregnancy. GHK-Cu's anti-fibrotic action is theoretically relevant to reducing pathological scar formation (keloid, hypertrophic scar) in the C-section incision line. No controlled trials have been reported.
Post-laser resurfacing recovery: A 2024 multicenter study investigated 0.05% GHK-Cu gel application after fractional CO₂ laser resurfacing — a procedure predominantly performed in women for photoaging. GHK-Cu gel accelerated re-epithelialization and reduced post-procedural erythema compared to standard wound care, consistent with earlier (2006, Archives of Facial Plastic Surgery) data demonstrating accelerated healing of CO₂-resurfaced skin with topical copper tripeptide complex. 35
9. Pulmonary Biology: COPD, Silicosis, and Acute Lung Injury
9.1 GHK-Cu and COPD: The Cigarette Smoke Study
Chronic obstructive pulmonary disease (COPD) affects over 300 million people globally. Its pathology involves emphysema (alveolar wall destruction from MMP and elastase activity) and chronic bronchitis (airway inflammation). GHK-Cu's documented ability to restore COPD fibroblast function was one of the first systemic effects identified by Pickart.
A Frontiers in Molecular Biosciences study (PubMed ID 35936787, 2022) directly characterized GHK-Cu's mechanism in a cigarette smoke-induced emphysema model: 36
- GHK-Cu attenuated emphysema by downregulating NF-κB pathway activation
- Simultaneously upregulated the Nrf2/Keap1 antioxidant pathway — the master transcriptional activator of phase II detoxification and antioxidant enzymes
- The convergence of NF-κB suppression and Nrf2 activation represents a dual-axis anti-inflammatory and antioxidant mechanism precisely suited to the oxidative-inflammatory pathology of smoke-induced lung damage
- At the protein level: decreased TNF-α, IL-6, and MCP-1; increased SOD and HO-1 (heme oxygenase-1)
9.2 Silicosis and PRDX6: A Novel Target Discovered in 2024
The 2024 Redox Biology study (PMC11228880) on GHK-Cu in silicosis employed a sophisticated target identification strategy to discover a previously unknown mechanism. 37
Experimental approach: Investigators used biotinylated GHK-Cu (GHK-Cu with a biotin tag attached) as molecular bait to capture its direct protein binding partners in RAW264.7 macrophages exposed to crystalline silica. Biotinylated immunoprecipitation followed by mass spectrometry proteome analysis identified Peroxiredoxin 6 (PRDX6) as a primary physical binding target.
The binding interaction was independently confirmed by:
- Biotinylated pull-down assays
- Western blot co-immunoprecipitation
- Surface plasmon resonance (SPR) — a biophysical technique that measures real-time molecular binding kinetics — revealing a binding affinity (Kd) of 2.81 × 10⁻⁵ M
Why PRDX6 matters: Peroxiredoxin 6 is a bifunctional enzyme — it acts as both a peroxidase (neutralizing H₂O₂ and lipid hydroperoxides) and a phospholipase A₂ (generating lysophospholipid signaling molecules). In macrophages exposed to crystalline silica, PRDX6 is a critical antioxidant defender; its suppression or damage accelerates silica-induced oxidative lung injury. By physically binding to and presumably stabilizing PRDX6, GHK-Cu exerts targeted antioxidant protection in macrophages at the site of silica-induced inflammation.
The specific Kd value (28 micromolar) is in a biologically plausible range for a peptide-protein interaction, consistent with the concentrations at which GHK-Cu produces in vivo effects.
In vivo outcomes: In the silicosis mouse model, GHK-Cu treatment:
- Attenuated silica-induced pneumonitis (lung inflammation) as assessed by histological scoring
- Reduced pulmonary fibrosis measured by hydroxyproline content (a collagen quantification marker)
- Reduced macrophage oxidative stress markers
- Produced no evidence of significant systemic toxicity (liver enzyme, kidney function markers were normal)
9.3 Acute Lung Injury
In lipopolysaccharide (LPS)-induced acute lung injury models, GHK-Cu:
- Prevented lung architectural disruption assessed by histology
- Suppressed inflammatory cell infiltration into alveolar spaces
- Increased SOD activity in lung tissue
- Decreased TNF-α and IL-6 through NF-κB p65 and p38 MAPK blockade
These findings are relevant to women because acute lung injury associated with severe infection, sepsis, or mechanical ventilation disproportionately produces lasting pulmonary fibrosis. 38
10. Bone Turnover, Menopause, and Skeletal Biology
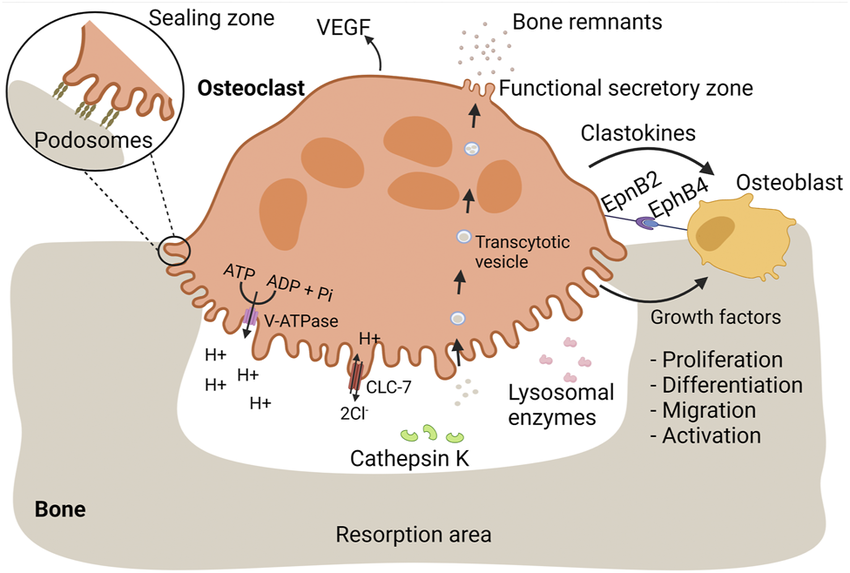
(https://commons.wikimedia.org/wiki/File:Osteoclasts_mediate_bone_resorption_and_proper_bone_replacement_by_osteoblasts.png) Figure 8: The bone remodeling cycle illustrating the coupled activities of osteoclasts (bone resorption, upper) and osteoblasts (bone formation, lower), regulated by the RANKL/OPG balance. Estrogen normally suppresses osteoclastogenesis by reducing RANKL and increasing OPG expression. GHK-Cu's documented NF-κB inhibition intersects this pathway: NF-κB activation is required for RANKL-induced osteoclast differentiation, providing a mechanistic basis for investigating GHK-type peptides in postmenopausal bone preservation. Source: Wikimedia Commons
10.1 The Postmenopausal Bone Loss Crisis
Postmenopausal women lose bone mineral density (BMD) at a rate of 1–3% per year in the first 5–7 years following menopause, driven by the removal of estrogen's inhibition of osteoclast (bone-resorbing cell) activity. Women lose a cumulative average of 35–40% of trabecular bone mass and 15–20% of cortical bone mass over their lifetime — rates approximately double those seen in age-matched men. Osteoporosis affects approximately 200 million women worldwide and is the primary driver of fragility fractures (hip, vertebral, wrist) that represent a leading cause of morbidity and mortality in women over 65. 39
The biological underpinning of this loss is the estrogen-RANKL axis: estrogen suppresses osteoblast expression of RANKL (receptor activator of NF-κB ligand), which stimulates osteoclast differentiation and activity. With estrogen withdrawal, RANKL expression rises, osteoclast activity increases, and the bone formation/resorption balance shifts toward net bone loss. NF-κB itself is central to RANKL signaling — osteoclastogenesis requires NF-κB activation in osteoclast precursors.
10.2 GHK-Cu's Mechanistic Relevance to Bone Biology
GHK-Cu has not been directly studied in postmenopausal osteoporosis RCTs with bone density endpoints. However, its documented mechanisms map onto bone biology in several ways:
NF-κB suppression and osteoclastogenesis: GHK-Cu's inhibition of NF-κB p65 phosphorylation may suppress RANKL-induced osteoclastogenesis — the same pathway targeted by denosumab (anti-RANKL antibody), a major approved osteoporosis treatment. Whether GHK-Cu produces meaningful osteoclast suppression at pharmacologically achievable concentrations in bone tissue remains unstudied.
Lysyl oxidase and bone collagen cross-linking: Bone matrix is ~90% type I collagen by organic mass. LOX-mediated cross-linking of bone collagen is essential for bone quality — not merely bone density. Poorly cross-linked bone is brittle regardless of mineral content. GHK-Cu's Cu²⁺ delivery to LOX supports collagen cross-linking quality in bone matrix, an effect not captured by DEXA scan (which measures mineral density, not collagen quality).
Anti-inflammatory cytokine suppression: TNF-α and IL-6 — both suppressed by GHK-Cu — are direct stimulators of RANKL expression and osteoclast differentiation. In post-menopausal women, elevated inflammatory cytokines drive an inflammatory component of bone loss (on top of the RANKL/estrogen mechanism) that GHK-Cu's anti-inflammatory profile theoretically addresses.
10.3 AHK-Cu Clinical Pilot: The Existing Human Data
The closest existing human evidence comes from a study of AHK-Cu (alanyl-histidyl-lysine copper) — a structural analog of GHK-Cu with very similar biological properties — in women at risk for osteoporosis, published in Biomedical Research (Biomedres.us). 40
The study investigated whether AHK-Cu patches worn by healthy aging women at risk for osteoporosis would produce measurable changes in bone turnover markers. Results:
- Significant changes in serum AHK-Cu levels and NTx (N-telopeptide) — a urinary marker of bone collagen resorption produced when osteoclasts degrade type I collagen cross-links
- Two participants demonstrated a slowing of bone loss and actual increase in bone density in previously compromised skeletal regions
- Authors concluded: "study data sufficiently significant to warrant further research" and called for adequately powered RCTs with DEXA scan endpoints
The NTx finding is mechanistically coherent: if GHK-type peptides suppress osteoclast activity (via NF-κB suppression), NTx — a direct osteoclast activity biomarker — would be expected to decrease. The limitation of this study is its small sample size and absence of a randomized control group; it functions as a hypothesis-generating pilot rather than efficacy evidence.
11. Reproductive Biology: Endometrial, Ovarian, and Fertility Research
11.1 Endometrial Biology and Implantation
The endometrium undergoes one of the most remarkable regenerative cycles in human biology — complete monthly reconstruction of the functional layer (stratum functionalis) from the basal layer under estrogen and progesterone control. Successful embryo implantation requires that the endometrium achieve a specific "receptive" state, characterized by:
- Adequate vascular density (spiral artery formation for embryo nutrient supply)
- ECM scaffold integrity (fibronectin, collagen IV, laminin in the basement membrane)
- Low inflammatory tone (paradoxically, a controlled pro-inflammatory state is required for implantation; excessive inflammation impairs it)
- Specific pinopode (surface projection) formation on the luminal epithelium
GHK-Cu's known mechanisms align with the first three requirements: VEGF-driven angiogenesis supports spiral artery formation; collagen IV upregulation supports basement membrane integrity; NF-κB modulation maintains controlled inflammatory tone. 41
Rodent evidence: Uterine injury models in rodents treated with GHK-Cu demonstrate: more rapid stromal tissue repair, thicker and more vascularized endometrium by histomorphometry, and in some models, improved reproductive outcomes (litter size) compared to untreated controls. These preclinical findings are internally coherent with GHK-Cu's known biology but do not directly translate to human endometrial receptivity without clinical data. 41
Limited human fertility data: Small pilot studies and case reports in assisted reproductive technology (ART) settings have observed increased endometrial thickness and successful embryo implantation following adjunctive local GHK-Cu administration. These observations are not from randomized controlled trials and carry high risk of confounding by concurrent IVF protocols. However, they provide the biological signal that motivates formal investigation.
11.2 Ovarian Biology: Granulosa Cell Protection
Granulosa cells (GCs) are the somatic cells of the ovarian follicle that directly support oocyte maturation by providing nutrients, hormones (estradiol, AMH), and growth signals. GC quality is a major determinant of oocyte competence and fertilization success.
Oxidative stress is a documented cause of GC dysfunction and apoptosis. In in vitro studies, GHK-Cu has been shown to reduce oxidative stress markers in granulosa cell lines — an effect mechanistically attributable to its upregulation of SOD and catalase activity in response to copper delivery and epigenetic antioxidant gene activation. 41
Separately, copper itself is an essential micronutrient for ovarian folliculogenesis. Copper deficiency impairs granulosa cell mitochondrial function and steroidogenesis. GHK-Cu's role as a copper chaperone may ensure adequate Cu²⁺ delivery to follicular tissue even when systemic copper status is suboptimal.
11.3 Anti-Müllerian Hormone Research Context
Anti-Müllerian hormone (AMH), produced by granulosa cells of small antral follicles, is the primary clinical biomarker of ovarian reserve. AMH levels decline with age, reflecting the progressive depletion of the primordial follicle pool. Oxidative stress accelerates this decline.
No direct GHK-Cu-AMH intervention studies have been published. However, given GHK-Cu's documented protection of granulosa cell oxidative status in vitro, an indirect mechanism through which sustained GHK-Cu exposure might slow AMH decline (by protecting granulosa cells that produce it from oxidative damage) is biologically plausible. This hypothesis is untested in clinical settings. 42
12. Cardiovascular Biology and Fibrinogen Suppression
12.1 The Fibrinogen Connection
GHK was originally identified in a specific context by Pickart: as a plasma fraction that suppressed fibrinogen synthesis in aged hepatocytes. Fibrinogen is a plasma glycoprotein produced by the liver that serves as the primary substrate for blood clot formation (via thrombin cleavage to fibrin). Chronically elevated fibrinogen is an independent cardiovascular risk factor — meta-analyses of prospective epidemiological studies have consistently found fibrinogen in the top tier of cardiovascular risk predictors alongside LDL-cholesterol and blood pressure. 1
Women's fibrinogen levels rise substantially after menopause — driven both by estrogen loss (estrogen suppresses fibrinogen synthesis via hepatic ERα signaling) and by rising inflammatory tone (fibrinogen is an acute-phase protein upregulated by IL-6). GHK-Cu's suppression of fibrinogen synthesis (direct) and IL-6 production (indirect, via NF-κB suppression) addresses fibrinogen elevation through both mechanisms.
12.2 Cardioprotection Against Copper Toxicity
A PMC study (PMC7564529, 2020) used zebrafish as an in vivo model to examine whether GHK-Cu could protect against cardiac toxicity from acute copper exposure. Findings: 43
- Recombinant GHK tripeptides protected zebrafish hearts from copper-induced cardiotoxicity
- The protective effect was observed at a GHK:Cu ratio of 1:10 — demonstrating that GHK's copper-chelating capacity absorbs excess ionic copper before it can produce cardiac oxidative damage
- Protection was confirmed by cardiac histology and functional parameters (heart rate regularity, morphological integrity)
The mechanism is direct copper chelation — GHK acting as a copper buffer that prevents ionic copper from engaging in Fenton chemistry in cardiac tissue. This cardioprotective role becomes relevant in the context of systemic copper dysregulation associated with aging and Alzheimer's disease.
13. Gastrointestinal and Hepatic Research
13.1 The 2025 Colitis Study: Full Methodological Detail
The most significant recent GHK-Cu gastrointestinal research is the 2025 Frontiers in Pharmacology study (PMC12263609, PubMed ID 40672369), which is worth examining in methodological depth because it established SIRT1 as a GHK-Cu target through a multi-tiered approach. 8
Animal model: Ulcerative colitis (UC) was induced in BALB/c mice using 3% dextran sulfate sodium (DSS) in drinking water for 14 days — the standard, reproducible murine UC model that produces colonic inflammation mimicking human UC histologically and symptomatically.
GHK-Cu intervention groups: DSS mice received GHK-Cu at varying doses vs. vehicle control; a positive control group received sulfasalazine (a standard UC treatment).
Outcome measures:
- Disease Activity Index (DAI): combining weight loss, stool consistency, and fecal bleeding scores — GHK-Cu alleviated weight loss and improved DAI vs. DSS controls
- Macroscopic: colon length (DSS causes colonic shortening from inflammation; GHK-Cu partially reversed this)
- Histology: inflammatory damage scoring, goblet cell enumeration (goblet cells produce protective mucus; their loss is characteristic of UC)
- Protein expression (Western blot): ZO-1, Occludin (tight junction proteins), TNF-α, IL-6, IL-1β, SIRT1, STAT3, p-STAT3, RORγt (Th17 marker)
In vitro corroboration: A co-culture system was constructed using mouse colonic epithelial cells (MCECs) + mouse peritoneal macrophages (MPMs) — an in vitro model capturing the epithelial-immune cell interaction central to mucosal barrier biology. GHK-Cu treatment of this co-culture model:
- Facilitated MCEC healing of DSS-impaired barrier
- Upregulated ZO-1 and Occludin (tight junction restoration — barrier repair)
- Upregulated SIRT1 protein
Target validation by gene silencing: To confirm that SIRT1 was the functionally relevant target (not merely a bystander), the investigators used STAT3 gene silencing by transfection in MCECs. With STAT3 knocked down, GHK-Cu's protective effects on tight junction proteins were attenuated — confirming that the SIRT1→STAT3 axis is mechanistically necessary for GHK-Cu's mucosal protective effects.
Network pharmacology and molecular docking: Prior to the animal experiments, the investigators used network pharmacology tools (protein interaction databases, target prediction algorithms) to identify SIRT1 as a top-ranked GHK-Cu target, then validated this computationally with molecular docking showing stable binding of GHK-Cu to the SIRT1 active site.
Significance for women: Ulcerative colitis affects women and men approximately equally, but disease course in women is significantly influenced by hormonal status. Estrogen has known anti-inflammatory effects in the gut; IBD flares are more common in perimenopausal women when estrogen levels fluctuate. GHK-Cu's SIRT1/STAT3-mediated mucosal protection represents an estrogen-independent pathway for gut anti-inflammatory support.
13.2 Historical Hepatic Evidence
Pickart's original work establishing GHK as a tissue-restorative signal used aged liver tissue as the primary model. GHK-Cu has been shown to:
- Restore aged hepatocyte function to a younger phenotype in vitro (the original discovery context)
- Protect liver tissue from oxidative damage in animal models
- Exhibit anti-fibrotic properties (TGF-β modulation) relevant to hepatic fibrosis progression
These hepatic effects have not been updated by large contemporary clinical studies, but the mechanistic groundwork is established. Non-alcoholic steatohepatitis (NASH) and its progression to fibrosis/cirrhosis represents a growing health crisis in postmenopausal women with metabolic syndrome — a population in which GHK-Cu's anti-fibrotic and antioxidant properties warrant formal investigation.
14. Cancer Biology: Tumor Suppressor Gene Modulation and Apoptosis Research
Important research context: The findings described in this section are derived from gene expression analyses and cell line studies. No clinical trials of GHK-Cu as a cancer treatment or cancer prevention agent have been conducted. These findings provide mechanistic hypothesis generation, not clinical guidance.
14.1 Gene Expression Analysis: The Anti-Cancer Profile
Analysis of GHK-Cu's gene expression signature against cancer-associated gene sets reveals a consistent anti-cancer transcriptional pattern. GHK suppresses RNA production in 70% of 54 human genes overexpressed in cancer patients — identified using the Broad Institute's LINCS database. 11
The suppressed genes include key oncogenic signaling hubs:
- YWHAB (14-3-3β): A scaffolding protein that protects oncogenic signaling complexes from degradation; its downregulation destabilizes cancer survival pathways
- MAP3K5 (ASK1): Context-dependent kinase; in cancer cells, promotes resistance to pro-apoptotic signals
- LMNA (Lamin A/C): Nuclear envelope protein mutated in multiple cancers; abnormal lamin expression contributes to nuclear deformability and invasive migration of cancer cells
- TGM2 (Transglutaminase 2): Promotes cancer cell survival, epithelial-mesenchymal transition (EMT), and chemotherapy resistance
14.2 Tumor Suppressor Upregulation
GHK-Cu simultaneously upregulates multiple tumor suppressor genes: 11
BRCA1: The most clinically significant finding — BRCA1 is a major DNA double-strand break repair gene whose loss-of-function mutations confer dramatically elevated lifetime risk of breast and ovarian cancer. GHK-Cu's upregulation of BRCA1 expression in non-cancer cells is consistent with its broader role in DNA repair gene activation (84 DNA repair genes upregulated). Whether this upregulation is physiologically meaningful (producing functional BRCA1 protein that participates in DNA repair) or merely reflects transcriptional noise requires further investigation.
PTEN: Phosphatase and tensin homolog; the most commonly mutated tumor suppressor in human cancer. PTEN opposes PI3K/AKT signaling — a pathway overactivated in breast, endometrial, and ovarian cancers.
TP73 (p73): A member of the p53 transcription factor family that induces apoptosis in response to DNA damage. p73 retains tumor-suppressive function in many cancers that have lost p53.
USP29: Deubiquitinase that stabilizes p53 protein by preventing its proteasomal degradation — effectively amplifying p53's tumor-suppressive activity. The upregulation of USP29 represents an indirect mechanism for GHK-Cu to enhance p53 function without directly altering TP53 gene expression.
LEFTY2: A TGF-β superfamily member whose overexpression in breast cancer cells has been shown to restrain tumor growth by inhibiting cancer cell invasiveness and epithelial-mesenchymal transition.
IL25 (IL-17E): A cytokine that induces apoptosis in tumor cells by activating caspase cascades via the TRAIL pathway.
14.3 Breast Cancer Cell Line Research
A study published in OBM Genetics examined GHK-Cu's modulation of gene expression in MCF7 human breast cancer cells (the most widely used estrogen receptor-positive breast cancer cell line) and PC3 prostate cancer cells. Key findings: 44
- GHK-Cu shifted MCF7 gene expression toward patterns more characteristic of normal, non-malignant mammary epithelium
- Upregulated caspase genes (CASP3, CASP7, CASP9) — executioner caspases that mediate apoptosis
- Upregulated genes in the apoptosis pathway that are typically silenced in cancer cells
- Gene set enrichment analysis showed downregulation of MYC target genes (a hallmark of aggressive breast cancer biology)
The MCF7 line is ER⁺ and progesterone receptor-positive (PR⁺) — the most common subtype of breast cancer in postmenopausal women. Whether GHK-Cu's gene modulation in MCF7 cells translates to reduced in vivo tumor growth requires animal model confirmation and ultimately clinical study.
14.4 DNA Repair: Genomic Integrity Maintenance
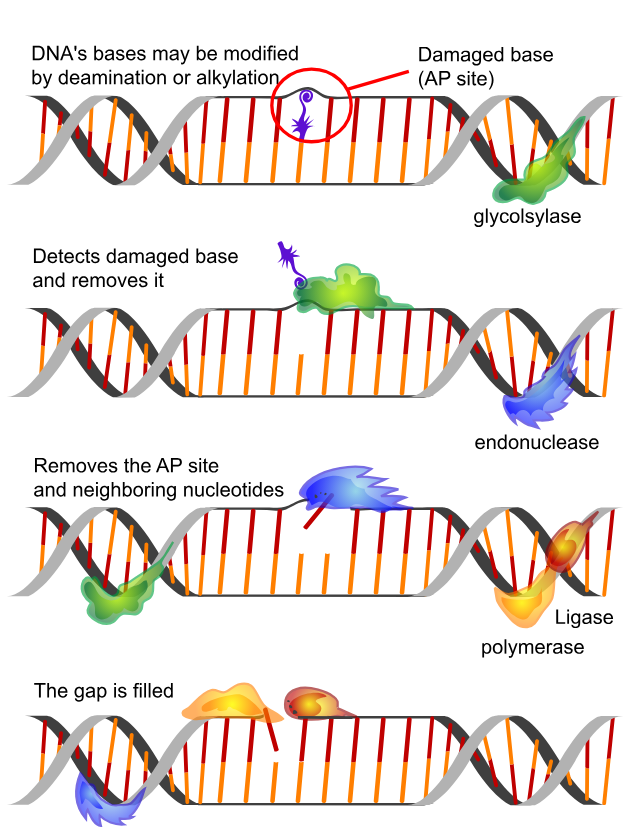
Figure 9: The base excision repair (BER) pathway — one of several DNA repair mechanisms upregulated in GHK-Cu genomic analyses. BER removes oxidatively damaged bases (e.g., 8-oxoguanine from ROS attack) through sequential glycosylase, AP endonuclease (APE1), DNA polymerase β, and DNA ligase activity. GHK-Cu's documented upregulation of 84 DNA repair genes includes components spanning BER, nucleotide excision repair (NER), mismatch repair (MMR), and double-strand break repair via homologous recombination — particularly relevant to BRCA1 stabilization. Source: Wikimedia Commons
GHK-Cu upregulates 84 genes associated with DNA repair processes: 10
- Base excision repair (BER): Corrects small base lesions from oxidative damage (8-oxoguanine) — the most common form of DNA damage in aging cells
- Nucleotide excision repair (NER): Corrects bulky lesions including those induced by UV radiation — directly relevant to skin cancer prevention
- Mismatch repair (MMR): Corrects replication errors; MMR gene mutations (MLH1, MSH2, MSH6) drive Lynch syndrome-associated colorectal and gynecological cancers
- Homologous recombination (HR): Repairs double-strand breaks; the pathway requiring BRCA1 and BRCA2
The coordinated upregulation of multiple repair pathways — rather than any single repair gene — is consistent with a broad epigenetic reprogramming of the cellular DNA damage response, not targeted induction of specific repair genes.
15. Epigenetics, DNA Repair, Stem Cell Biology, and Aging Clocks
15.1 The Epigenetic Reset Hypothesis
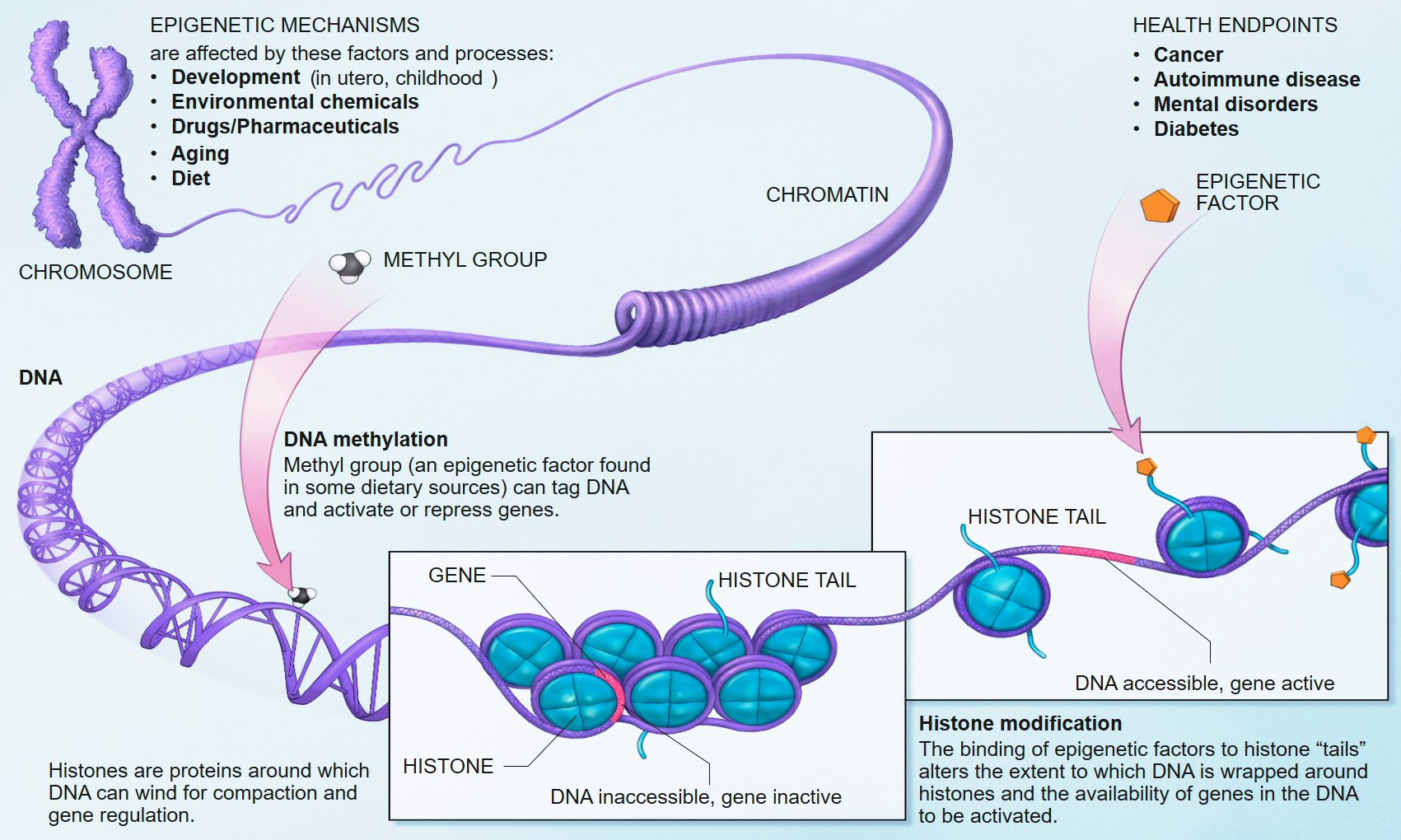
Figure 10: Epigenetic regulatory mechanisms modulating gene expression without altering DNA sequence. DNA methylation (CpG island methylation silences gene promoters), histone modifications (acetylation/deacetylation, methylation/demethylation, phosphorylation), and chromatin remodeling collectively determine which genes are accessible for transcription. GHK-Cu's proposed epigenetic reset function — restoring methylation patterns at regenerative gene promoters toward a younger state — operates through these mechanisms, particularly SIRT1-mediated histone deacetylation and indirect modulation of DNA methyltransferase activity. Source: National Institutes of Health / Wikimedia Commons
Pickart and Margolina's 2014 paper "GHK and DNA: Resetting the Human Genome to Health" (BioMed Research International, PMC4180391) advanced the hypothesis that GHK-Cu functions as an epigenetic reset signal — reversing the accumulation of methylation changes at regenerative gene promoters that characterizes cellular aging. 10
The theoretical basis is as follows: aging is accompanied by progressive CpG island methylation at promoters of genes encoding growth factors, tissue repair proteins, and stem cell factors — silencing these genes even though the DNA sequence encoding them remains intact. If GHK-Cu can induce demethylation or chromatin remodeling at these promoters, it could reactivate silenced regenerative programs without altering the genome itself.
The 2024 EurekAlert-reported clinical trial providing direct evidence for this mechanism in skin tissue used gene expression profiling of biopsied skin to demonstrate that GHK-Cu gel application activated epigenetic regulatory pathway genes — chromatin remodeling enzymes (histone acetyltransferases, demethylases) — in skin fibroblasts in vivo, not merely the downstream collagen-synthesis genes they control. This represents mechanistic evidence in human tissue, not just in vitro speculation. 20
15.2 Biological Age Clocks and GHK-Cu
Epigenetic clocks — mathematical models based on DNA methylation patterns at specific CpG sites (Horvath clock, GrimAge, PhenoAge) — have emerged as the most accurate biological age estimators, often diverging from chronological age to reflect accelerated or decelerated aging. These clocks directly measure the methylation state at genomic loci associated with aging.
If GHK-Cu produces widespread epigenetic changes (as the gene expression and mechanism data suggest), it could theoretically shift epigenetic clock readings toward a younger biological age. This hypothesis has not been directly tested in published clinical studies as of April 2026 — representing a significant methodological opportunity for future research, as epigenetic clock analyses could be added to ongoing clinical trials at minimal additional cost.
15.3 Stem Cell Biology: GHK-Cu and MSC Function
A study published in OBM Geriatrics (Lidsen, 2018) examined GHK-Cu's effects on mesenchymal stem cells (MSCs) — the progenitor cells responsible for generating osteoblasts, chondrocytes, adipocytes, and fibroblasts. 45
Key findings:
- GHK-Cu increases MSC secretion of hepatocyte growth factor (HGF), epidermal growth factor (EGF), and fibroblast growth factor (FGF) — trophic factors that support tissue repair and stem cell homing
- Upregulates gene expression of stemness markers associated with MSC self-renewal capacity
- Supports the cellular stemness phenotype — the capacity for cells to undergo self-renewal and multipotent differentiation
As MSC populations decline with age and are depleted by repeated tissue injury, stress, and inflammation, maintaining MSC secretory function becomes increasingly important for systemic tissue maintenance. GHK-Cu's support of MSC trophic factor production represents a systemic regenerative mechanism that extends beyond any individual tissue.
15.4 Mitochondrial Biology
GHK-Cu's relevance to mitochondrial function extends beyond copper delivery to Complex IV. The April 2026 bioRxiv study identified sustained suppression of mitochondrial metabolic signaling in intranasally-treated hippocampi — a gene expression pattern associated with caloric restriction-induced longevity and with the suppression of the mTOR pathway that governs cellular growth and metabolic rate. 30
Mitochondrial DNA is particularly vulnerable to oxidative damage because:
- It lacks the protective histone-chromatin packaging of nuclear DNA
- It has limited repair mechanisms
- It is adjacent to the primary intracellular ROS source (the electron transport chain)
GHK-Cu's upregulation of SOD2 (mitochondrial superoxide dismutase) specifically addresses the mitochondrial oxidative burden — SOD2 is the primary defense against superoxide generated at Complexes I and III.
16. Bioavailability, Pharmacokinetics, and Delivery System Research
16.1 Topical Penetration: The Stratum Corneum Problem
GHK-Cu is a hydrophilic molecule (logP negative, high water solubility). The stratum corneum — the outermost layer of skin — is a lipophilic barrier (composed of corneocytes embedded in a lipid matrix rich in ceramides, cholesterol, and fatty acids) that preferentially excludes hydrophilic molecules. This creates a fundamental bioavailability challenge for topical GHK-Cu application.
A 2025 study (PMC11721469) specifically addressed the question: Are existing methods adequate to measure skin permeation of GHK-Cu encapsulated in liposomes? Key methodological findings: 46
- The study found that standard Franz diffusion cell methods using synthetic membranes significantly underestimate GHK-Cu penetration into real skin tissue
- Measurement at multiple skin layers (stratum corneum, viable epidermis, dermis separately) is necessary for accurate bioavailability characterization
- Liposomal encapsulation substantially improved peptide delivery to viable epidermis and dermis compared to aqueous solution — confirming that delivery formulation is as important as active ingredient concentration
Clinical implication of the penetration data: Commercial GHK-Cu serums at 0.05–1% may deliver less active peptide to dermal fibroblasts than formulation concentration alone would suggest. The strong clinical results seen with nanomolar concentrations in in vitro studies could potentially be achieved topically if delivery systems reliably reach the dermis — a gap that nanoliposomal, ionic liquid, and microemulsion delivery technologies are now specifically addressing.
16.2 Ionic Liquid Microemulsions: The 2024 Delivery Breakthrough
The ionic liquid microemulsion approach (PMC10643103, 2024) employs room-temperature ionic liquids — salts that are liquid at ambient temperature — as delivery vehicles. Ionic liquids can solubilize both hydrophilic and lipophilic components, creating thermodynamically stable microemulsions that penetrate the stratum corneum more effectively than conventional aqueous vehicles.
The study demonstrated approximately threefold increase in local follicular delivery of copper peptides compared to aqueous solution — quantified by fluorescence imaging of labeled peptide distribution in ex vivo scalp tissue. The threefold improvement translates the nanomolar effective concentrations established in vitro into a pharmacologically achievable range for topical clinical application. 27
16.3 Subcutaneous Injection Pharmacokinetics
Subcutaneous and intramuscular injection provide direct systemic bioavailability (near 100% for non-protein-bound fraction). The primary pharmacokinetic limitation is GHK-Cu's short plasma half-life — estimated at under 60 minutes — consistent with rapid renal clearance of a small, non-protein-bound peptide and uptake by target tissues. 47
This short half-life means that intermittent injections produce pulsatile rather than sustained plasma GHK-Cu elevation. Whether pulsatile or sustained delivery is more effective for specific biological targets is not well characterized. The bioRxiv 2026 study's finding of divergent effects between 5-day IP (pulsatile, short-term) and 8-week IN (sustained, long-term) GHK-Cu exposure is directly relevant to this pharmacokinetic question.
16.4 Intranasal Delivery and CNS Access
The detection of GHK in cerebrospinal fluid raises the question of how GHK-Cu reaches the CNS. Two mechanisms are plausible:
-
Passive diffusion across the BBB: At MW ~404 Da, GHK-Cu is within the size range (generally <500 Da) often considered permissive for passive BBB crossing. However, its hydrophilicity would reduce passive transcellular diffusion.
-
Direct olfactory/trigeminal pathway from intranasal administration: Intranasal delivery allows peptides to bypass the BBB entirely by traveling along olfactory and trigeminal nerve pathways from the nasal epithelium directly to brain parenchyma. The bioRxiv 2026 study's finding that intranasal delivery produces markedly different (and more sustained) hippocampal gene expression changes than systemic IP delivery is consistent with direct CNS delivery via olfactory pathways producing distinct pharmacological effects compared to systemic delivery reaching the brain via the BBB. 30
This has major implications for designing human neuroprotection studies — intranasal delivery may be mechanistically necessary for the specific hippocampal aging-suppression program observed in the 2026 study.
17. Safety Pharmacology and Toxicology
17.1 Topical Safety Record
GHK-Cu has the most extensive safety record in its topical form, having been incorporated in cosmetic formulations for over three decades. Published studies consistently report:
- No carcinogenicity — in fact, the gene expression profile shows anti-proliferative, pro-apoptotic effects in cancer cells
- No mutagenicity in Ames test models
- Most common adverse effect: mild, transient erythema at higher concentrations (2–3%), particularly on sensitive skin or in initial use — mechanistically attributed to copper ion stimulation of local vasodilation rather than allergic reaction
- Contact dermatitis to GHK-Cu itself is rare; reactions more commonly attributable to formulation excipients 47
17.2 Systemic Safety and Dose-Effect Relationships
At very high doses, GHK-Cu has been associated with blood pressure reduction in animal models — proposed to occur via copper-mediated activation of eNOS (endothelial nitric oxide synthase), which generates nitric oxide (a potent vasodilator). This effect has not been observed at therapeutic doses in human studies, but it represents a theoretical consideration for individuals with preexisting hypotension or on vasodilatory medications.
Published research reports no serious adverse events in studies using doses up to 3 mg daily subcutaneously for periods of several months. 47
17.3 Pregnancy and Lactation
No teratogenicity studies for GHK-Cu have been reported in peer-reviewed literature as of April 2026. The absence of safety data in pregnant and lactating women — not demonstrated harm — is the basis for caution in these populations. Given that copper metabolism is extensively altered during pregnancy (copper requirements increase substantially due to fetal neurological development), and that GHK-Cu acts as a copper chaperone, the theoretical interaction between exogenous GHK-Cu and maternal-fetal copper homeostasis has not been characterized.
17.4 Drug Interaction Profile
No pharmacokinetic drug-drug interactions have been formally studied. Theoretical interactions of mechanistic concern:
- Copper chelators (D-penicillamine, trientine, tetrathiomolybdate): Used in Wilson's disease (copper overload disorder); would compete with GHK for copper and reduce GHK-Cu complex formation
- High-dose zinc supplementation (>25 mg/day): Zinc and copper compete for intestinal absorption via metallothionein; chronic high zinc intake reduces copper bioavailability and could reduce endogenous GHK-Cu formation
- Systemic corticosteroids: Cortisone inhibits wound healing; GHK-Cu has been shown in animal studies to partially overcome cortisone's inhibitory effect on wound repair — a potentially beneficial interaction 1
18. Age-Stratified Biology: What Research Reveals About GHK-Cu Across Women's Life Stages
This section synthesizes what the research literature reveals about the biological state of GHK-Cu signaling and documented tissue responses across women's life stages. This is a research-findings synthesis, not a clinical protocol.
18.1 The Second and Early Third Decade (Ages 20–30): Peak GHK-Cu Signaling Biology
Endogenous GHK-Cu biology at this stage: Plasma GHK levels are at or near their lifetime peak (~200 ng/mL). Fibroblast responsiveness to GHK-Cu in culture is at its highest — studies comparing fibroblasts isolated from donors of different ages consistently find that young-adult fibroblasts show greater collagen synthetic response to GHK-Cu stimulation than aged fibroblasts. 16 This age-dependent responsiveness difference is believed to reflect the progressive silencing of GHK-responsive gene networks in aged fibroblasts — epigenetic changes that dampen the cell's capacity to respond to the peptide signal even when the signal is present.
Hair follicle biology: Anagen phase duration is at maximum (~6 years for scalp follicles), follicle diameter is at its lifetime peak, and Wnt/β-catenin signaling is robustly active. The dermal papilla microenvironment is richly vascularized and collagen-organized. In this context, GHK-Cu's follicle-biology effects are studied primarily in relation to accelerated miniaturization from androgen excess (PCOS-associated FPHL) or nutritional/inflammatory-driven telogen effluvium — contexts where GHK-Cu's antioxidant and Wnt-reactivating properties may be relevant regardless of the high background GHK-Cu milieu.
Wound healing: The hallmark of healing biology in this decade is speed and completeness. The fibroblast proliferation, collagen synthesis, and angiogenesis responses operate at maximum efficiency. Scar quality, however — not speed — is the primary limitation, as younger skin has higher TGF-β1 activity that promotes scarring. GHK-Cu's TGF-β modulating and anti-fibrotic properties are most studied in this context (acne scars, surgical scars) in young-adult skin.
Research gap at this life stage: Almost no GHK-Cu clinical research has specifically enrolled women in their 20s. Study populations are predominantly 40+ years, leaving the biology at peak GHK-Cu levels largely uncharacterized in formal trials.
18.2 The Third and Fourth Decade (Ages 30–45): The Descending Phase
Endogenous biology: Plasma GHK levels decline from ~200 ng/mL toward ~130–150 ng/mL across this decade — a 25–35% reduction. Concurrently, collagen synthesis rates begin declining measurably (approximately 1% per year in skin fibroblasts after age 25 — documented by biopsy studies), and early perimenopause biochemical changes (rising FSH, declining AMH, estrogen fluctuation) begin affecting tissue biology even before clinical symptoms are apparent.
Hair biology: FPHL can present in women as young as 25–30, particularly in those with PCOS, insulin resistance, or family history. The follicle miniaturization process, once established, is progressive and difficult to reverse — making this decade a critical window for biological research on follicle preservation. Studies on GHK-Cu in the 30–45 age group are underrepresented; most hair loss trials enroll women with established FPHL (typically >40 years). The 2025 minoxidil-dutasteride-copper peptide tattooing study (PMC11992372) did not report age-stratified outcomes. 26
Reproductive biology: This is the primary decade of fertility medicine relevance for GHK-Cu. Research documenting GHK-Cu's effects on endometrial vascularity and granulosa cell oxidative stress (as described in Section 11) is most clinically relevant here, as women seeking fertility treatment (IVF, IUI) are predominantly in this age range. The absence of RCTs specifically examining GHK-Cu in ART populations is a significant research gap given the biological plausibility established in rodent models and in vitro studies.
Postpartum biology: The postpartum period involves dramatic hormonal shifts (estrogen and progesterone collapse within 24 hours of delivery), extensive tissue repair (uterine involution, perineal healing, breast tissue remodeling), and frequently telogen effluvium at 2–4 months postpartum. GHK-Cu's relevant mechanisms (angiogenesis, collagen synthesis, follicle Wnt reactivation) are all engaged in postpartum biology, but no studies have examined GHK-Cu specifically in postpartum women.
18.3 The Perimenopause Decade (Ages 45–55): Convergent Decline
Endogenous biology: This decade represents the convergence of GHK-Cu decline (~40–50% below peak) with the estrogen collapse of menopause. As detailed in Section 4, both declines are mechanistically significant — and they converge on the same biological endpoints (collagen, VEGF, NF-κB regulation, BDNF). The compounded biological impact explains why the rate of visible aging accelerates in perimenopausal women and why this is the decade most investigated in GHK-Cu clinical trials.
Skin biology research at this stage: The majority of GHK-Cu clinical trials (the 71-woman photoaging study, the 60-woman split-face RCT, the 21-woman epigenetic trial, the comparative collagen study vs. vitamin C/retinoic acid) enrolled women primarily in the 40–65 age range. The accumulated data establishes that this is the life stage where topical GHK-Cu produces clinically measurable collagen density improvements. The 2024 epigenetic trial's finding that GHK-Cu activates chromatin remodeling genes in vivo in this population suggests the aging-associated epigenetic silencing of collagen genes is not irreversible — it can be partially overcome by GHK-Cu-mediated epigenetic intervention. 20
Cognitive biology at this stage: Neuroimaging studies demonstrate hippocampal volume loss and reduced BDNF protein levels in women entering perimenopause compared to premenopausal controls. The subjective "brain fog" reported by 60–70% of perimenopausal women has objective neural correlates in working memory task performance and PET imaging of cerebral glucose metabolism. GHK-Cu's documented upregulation of BDNF and NGF genes, and its NF-κB-mediated neuroinflammation suppression, are mechanistically coherent with the biology of this window — yet no clinical trial has examined GHK-Cu's cognitive effects specifically in perimenopausal women. This is the most significant research gap in GHK-Cu's women's health profile.
Bone biology: The first 5 years of postmenopause represent peak bone loss rate. Research suggests that bone loss detectable biochemically (rising NTx) begins 2–5 years before the final menstrual period, during late perimenopause. The AHK-Cu pilot showing changes in NTx enrolled women at risk for osteoporosis — likely predominantly in this age range. 40
18.4 The Postmenopause Phase (Ages 55–70): Established Deficiency
Endogenous biology: Plasma GHK-Cu at ~80 ng/mL represents a sustained 60% deficit below peak. Concurrent with this, estrogen is near-absent, systemic inflammatory markers (CRP, IL-6, fibrinogen) are elevated, bone loss continues (though more slowly than in the first postmenopausal decade), and Alzheimer's disease risk begins rising steeply. Women in this phase have lost multiple parallel systems (GHK-Cu signaling, estrogen signaling) that maintained tissue homeostasis.
Brain biology: The April 2026 bioRxiv study enrolled mice at 20–21 months — equivalent to approximately 60–65 human years — and demonstrated that GHK-Cu produces measurable improvement in hippocampal-dependent cognition at this age. The intranasal route's superior effect on hippocampal aging gene programs is mechanistically relevant to this demographic: the sustained suppression of mitochondrial aging metabolic signaling (associated with the CR-mimetic/mTOR-suppressive molecular program) may slow the progression of age-related hippocampal synaptic loss. 30
Gut and metabolic biology: SIRT1 activity declines with age across multiple tissues. GHK-Cu's establishment as a SIRT1 activator (2025 colitis study) is most consequential at life stages where SIRT1 activity is most depressed. Postmenopausal women with metabolic syndrome have particularly low SIRT1 activity in metabolic tissues (liver, adipose, gut), and the SIRT1/STAT3 axis identified in the colitis study may have relevance in NASH, metabolic inflammation, and gut barrier dysfunction in this population.
Cancer biology relevance: The incidence of breast cancer peaks in postmenopausal women aged 60–70. GHK-Cu's gene expression effects on BRCA1 expression, p53 stabilization (via USP29 upregulation), and MCF7 cell transcriptome normalization are most biologically relevant at this life stage where accumulation of mutational load and reduced DNA repair capacity create peak cancer vulnerability. The research does not yet establish protective efficacy but provides a mechanistic framework. 11
18.5 The Late Life Phase (Ages 70+): Maximum Biological Need, Minimum Responsiveness
Endogenous biology: GHK-Cu levels are at or below 80 ng/mL. Crucially, research suggests that fibroblast responsiveness to GHK-Cu also declines with age — older fibroblasts show reduced collagen synthetic response to GHK stimulation compared to young fibroblasts, even when GHK-Cu is supplied at equivalent concentrations. This phenomenon is believed to reflect the progressive epigenetic silencing of GHK-responsive gene networks across the lifespan. 16
This creates a biological paradox: the population with the greatest need for GHK-Cu signaling (most depleted endogenously, most tissue degradation) is also the population least capable of responding to it. The 2024 epigenetic clinical trial's finding that GHK-Cu activates chromatin remodeling enzymes (not merely downstream collagen genes) suggests a mechanistic pathway through which GHK-Cu might restore responsiveness by reversing the epigenetic silencing — but whether this works equally in 70-year-old fibroblasts as in 50-year-old fibroblasts is unstudied.
Wound healing: Delayed wound healing in elderly women is a major source of morbidity — hip fracture surgical wounds, diabetic foot ulcers, pressure ulcers. The preclinical evidence showing 64.5% wound area reduction in GHK-Cu-treated rodent models is most clinically pressing in this population, yet clinical trials in elderly women's wound healing have not been reported.
Stem cell biology: Mesenchymal stem cell populations decline with age, and those that remain show reduced proliferative and differentiation capacity. GHK-Cu's documented support of MSC trophic factor secretion (HGF, EGF, FGF) represents a mechanism to enhance the functional output of a depleted stem cell population without necessarily expanding cell numbers — analogous to improving the efficiency of remaining machinery rather than adding new machinery.
19. Research Gaps and Future Directions
A rigorous review of GHK-Cu literature reveals that while mechanistic and preclinical evidence is extensive, the clinical trial base specific to women is sparse and poorly age-stratified. The following gaps represent the highest-priority research opportunities:
Perimenopausal cognitive health: No prospective RCT has examined GHK-Cu's effects on cognitive function in perimenopausal or early postmenopausal women — despite compelling mechanistic rationale (BDNF upregulation, NF-κB neuroinflammation suppression) and an established model in aged rodents showing cognitive improvement via intranasal delivery. A 12-week RCT in perimenopausal women with subjective cognitive complaints, using validated cognitive instruments and optional BDNF plasma measurement, would be highly informative.
Postmenopausal osteoporosis: The AHK-Cu pilot provides an NTx signal but no DEXA-based BMD data. A 12–24 month RCT in postmenopausal women not on pharmacological osteoporosis treatment, with DEXA endpoints and bone turnover marker (CTX, P1NP, NTx) secondary outcomes, would fill the most clinically significant gap in GHK-Cu bone research.
Epigenetic clock validation: Given the 2024 clinical evidence of epigenetic mechanism activation in skin, adding epigenetic clock analysis (GrimAge or DunedinPACE) to existing or future GHK-Cu trials would test the hypothesis that GHK-Cu reduces biological age as measured by the methylome — a direct test of the most theoretically significant longevity claim.
Fertility and ART: A placebo-controlled RCT of topical or intrauterine GHK-Cu in women undergoing IVF with thin endometrium or poor previous implantation, measuring endometrial thickness, pinopode formation, and live birth rate, would address the most clinically urgent reproductive research gap.
Intranasal delivery in humans: The April 2026 bioRxiv study's finding that intranasal GHK-Cu produces distinct and superior hippocampal aging-suppression programs compared to systemic delivery motivates a first-in-human intranasal GHK-Cu trial with cognitive and biofluid BDNF outcomes.
FPHL in women, age-stratified: A head-to-head comparison of GHK-Cu scalp application vs. topical minoxidil in premenopausal women with FPHL, with trichoscopic endpoint (follicle density, diameter) and standardized photography, with sex hormone and AMH stratification, would clarify GHK-Cu's clinical positioning in women's hair loss.
Postpartum telogen effluvium: A small RCT in postpartum women with clinically documented telogen effluvium (hair shedding >100/day at 2–4 months postpartum), using GHK-Cu scalp serum vs. vehicle, with trichogram endpoint at 3 months, would test the Wnt-reactivation hypothesis in a well-defined biological context.
Long-term Phase 3 safety: All existing injectable GHK-Cu studies are short-term (<6 months). A long-term safety study (12–24 months) with standardized adverse event reporting, serum copper monitoring, and liver/kidney function panels is necessary before broader clinical adoption.
20. Complete Source Index
Compiled from peer-reviewed journals, preprint repositories, and registered clinical study data through April 2026. All images linked to original host sources. Source access dates: March–April 2026.
Footnotes
-
Pickart L. The human tri-peptide GHK and tissue remodeling. Journal of Biomaterials Science, Polymer Edition. 2008. Original GHK discovery and endogenous biology. https://pmc.ncbi.nlm.nih.gov/articles/PMC8789089/ ↩ ↩2 ↩3 ↩4 ↩5
-
GHK-Cu Peptide | The benefits, side effects, and more [2026]. Innerbody Research. Copper binding affinity pKa values. https://www.innerbody.com/ghk-cu-peptide ↩ ↩2 ↩3
-
GHK-Cu Peptide Explained: Structure, Mechanism, and Research Insights. BioPrime USA. Copper coordination chemistry. https://bioprimeusa.com/ghk-cu-peptide-explained-structure-mechanism-and-research-insights/ ↩ ↩2 ↩3 ↩4
-
Expression of Glycosaminoglycans and Small Proteoglycans in Wounds: Modulation by Glycyl-L-Histidyl-L-Lysine-Cu2+. Journal of Investigative Dermatology (1992). Lysyl oxidase and ECM organization. https://www.sciencedirect.com/science/article/pii/S0022202X1541067X ↩
-
GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration. PMC (2015). PMC4508379. SOD activation, antioxidant mechanisms. https://pmc.ncbi.nlm.nih.gov/articles/PMC4508379/ ↩ ↩2 ↩3
-
MAPKs and NF-κB signaling pathway in the anti-inflammatory effect of GHK-Cu. ResearchGate (Figure from LPS/RAW264.7 study). p38 MAPK, JNK, NF-κB p65 phosphorylation data. https://www.researchgate.net/figure/MAPKs-and-NF-kB-signaling-pathway-is-involved-in-the-anti-inflammatory-effect-of-GHK-Cu_fig7_306042505 ↩ ↩2
-
GHK-Cu Research: Complete Scientific Guide to Copper Peptide. Oath Peptides (2025). SIRT1 molecular docking, -8.75 kcal/mol binding energy. https://oathpeptides.com/2025/11/18/ghk-cu-research-complete-scientific-guide-to-copper-peptide/ ↩ ↩2
-
Exploring the beneficial effects of GHK-Cu on an experimental model of colitis and the underlying mechanisms. Frontiers in Pharmacology (2025). PMC12263609. SIRT1/STAT3 pathway, DSS colitis methodology, ZO-1/Occludin findings. https://pmc.ncbi.nlm.nih.gov/articles/PMC12263609/ ↩ ↩2
-
Exploring the Role of Tripeptides in Wound Healing and Skin Regeneration: A Comprehensive Review. MedSci (2025). TGF-β modulation, ECM proteoglycans, elastin. https://www.medsci.org/v22p4175.htm ↩ ↩2 ↩3 ↩4 ↩5 ↩6
-
GHK and DNA: Resetting the Human Genome to Health. BioMed Research International (2014). PMC4180391. 4,000+ genes, CMap analysis, 31.2% genome modulation, DNA repair genes, epigenetic reset hypothesis. https://pmc.ncbi.nlm.nih.gov/articles/PMC4180391/ ↩ ↩2 ↩3 ↩4 ↩5 ↩6
-
GHK, the Human Skin Remodeling Peptide, Induces Anti-Cancer Expression of Numerous Caspase, Growth Regulatory, and DNA Repair Genes. Journal of Analytical Oncology (2015). BRCA1, PTEN, TP73, USP29, LEFTY2, IL25, YWHAB, MAP3K5 findings. https://neoplasiaresearch.com/pms/index.php/jao/article/view/217 ↩ ↩2 ↩3 ↩4
-
Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. PMC (2018). PMC6073405. Gene categories table, fibrinogen suppression data. https://pmc.ncbi.nlm.nih.gov/articles/PMC6073405/ ↩
-
Skin collagen changes in postmenopausal women receiving different regimens of estrogen therapy. PubMed (1987). PMID 3601260. 30% collagen loss in first 5 years postmenopause finding, HRT collagen restoration. https://pubmed.ncbi.nlm.nih.gov/3601260/ ↩ ↩2
-
Reducing the Diseases of Human Aging with GHK-Cu: Reversing the Effects of Menopause on Skin. SkinBio.com. 70% vs. 50% vs. 40% collagen improvement comparison study. http://skinbio.com/menopause.php ↩ ↩2
-
Effects of Copper Tripeptide on the Growth and Expression of Growth Factors by Normal and Irradiated Fibroblasts. Archives of Facial Plastic Surgery (2005). VEGF and FGF-2 upregulation at 1 nM GHK-Cu. https://www.liebertpub.com/doi/10.1001/archfaci.7.1.27 ↩ ↩2 ↩3
-
Stimulation of collagen synthesis in fibroblast cultures by the tripeptide-copper complex glycyl-L-histidyl-L-lysine-Cu2+. PubMed (1988). PMID 3169264. Biphasic dose-response, 1–10 nM optimal concentration, 50–200% synthesis increase. https://pubmed.ncbi.nlm.nih.gov/3169264/ ↩ ↩2 ↩3
-
Synergy of GHK-Cu and hyaluronic acid on collagen IV upregulation via fibroblast and ex-vivo skin tests. Journal of Cosmetic Dermatology (2023). PMID 37062921. Collagen I, IV, VII qRT-PCR, ex vivo immunofluorescence, 0.3 mg/mL total concentration. https://pubmed.ncbi.nlm.nih.gov/37062921/ ↩
-
GHK Cu Peptide | Clinical Evidence & Skin Benefits 2025. Grand Ingredients. 71-woman photoaging study detail. https://grandingredients.com/copper-peptides-clinical-benefits/ ↩
-
GHK-Cu Research Results: What Preclinical and Clinical Studies Show in 2026. Spartan Peptides. 60-woman split-face RCT (22% firmness, 16% fine line reduction); nano-lipid carrier study (55.8%, 31.6%, 32.8% wrinkle outcomes). https://spartanpeptides.com/blog/ghk-cu-research-results-preclinical-clinical-studies-2026/ ↩ ↩2
-
Epigenetic mechanisms activated by GHK-Cu increase skin collagen density in clinical trial. EurekAlert! (2024). 21-woman trial; 28% mean collagen increase, 51% top quartile; epigenetic regulatory pathway gene activation in biopsy specimens. https://www.eurekalert.org/news-releases/990464 ↩ ↩2 ↩3
-
Advances in Transdermal Delivery Systems for Treating Androgenetic Alopecia. Pharmaceutics (2025). FPHL epidemiology, follicle miniaturization biology. https://www.mdpi.com/1999-4923/17/8/984 ↩ ↩2
-
The effect of tripeptide-copper complex on human hair growth in vitro. ResearchGate (2006). Human hair follicle organ culture, follicle elongation and diameter increase. https://www.researchgate.net/publication/6135527 ↩
-
The Molecular Mechanism of Natural Products Activating Wnt/β-Catenin Signaling Pathway for Improving Hair Loss. PMC (2022). PMC9693075. Wnt/β-catenin mechanism in hair follicle cycling; TCF/LEF transcription. https://pmc.ncbi.nlm.nih.gov/articles/PMC9693075/ ↩
-
Behavioral and neuropathological features of Alzheimer's disease are attenuated in 5xFAD mice treated with intranasal GHK peptide. PMC (2023). PMC10690187. Amyloid plaque reduction, microglial activation, SOD/catalase normalization, behavioral improvement. https://pmc.ncbi.nlm.nih.gov/articles/PMC10690187/ ↩ ↩2
-
Randomized controlled trial on a PRP-like cosmetic, biomimetic peptides based, for the treatment of alopecia areata. Journal of Dermatological Treatment (2018). Hair density score improvement, RCT design. https://www.tandfonline.com/doi/full/10.1080/09546634.2018.1544405 ↩
-
Enhanced hair regrowth with five monthly sessions of minoxidil-dutasteride-copper peptides tattooing for androgenetic alopecia assessed by artificial intelligence and blinded evaluators. PMC (2025). PMC11992372. https://pmc.ncbi.nlm.nih.gov/articles/PMC11992372/ ↩ ↩2
-
Thermodynamically stable ionic liquid microemulsions pioneer pathways for topical delivery and peptide application. PMC (2023/2024). PMC10643103. Threefold follicular delivery improvement data. https://pmc.ncbi.nlm.nih.gov/articles/PMC10643103/ ↩ ↩2
-
GHK-Cu and Neuroprotection: Research on Copper Peptide Brain and Nerve Effects. Spartan Peptides. Surgical menopause cognitive acceleration, estrogen-BDNF axis summary. https://spartanpeptides.com/blog/ghk-cu-neuroprotection-brain-nerve-regeneration-research/ ↩
-
The Effect of the Human Peptide GHK on Gene Expression Relevant to Nervous System Function and Cognitive Decline. Brain Sciences (2017). PMC5332963. NGF, BDNF, NGFR upregulation; BACE1 downregulation; presenilin gene modulation. https://pmc.ncbi.nlm.nih.gov/articles/PMC5332963/ ↩
-
Middle-aged mice treated with GHK-Cu peptide administered intraperitoneally or intranasally show behavioral rescue but divergent hippocampal aging programs. bioRxiv (April 13, 2026). DOI: 10.64898/2026.04.09.717524v1. IP vs. IN route divergence; NES values for oxidative phosphorylation, DNA repair, MYC targets; transcriptomic analysis. https://www.biorxiv.org/content/10.64898/2026.04.09.717524v1 ↩ ↩2 ↩3 ↩4
-
A Copper-Binding Peptide with Therapeutic Potential against Alzheimer's Disease: From the Blood-Brain Barrier to Metal Competition. PubMed (January 2025). PMID 39723808. Copper-amyloid-beta interaction mechanism; BBB permeability characterization. https://pubmed.ncbi.nlm.nih.gov/39723808/ ↩
-
Does GHK-Cu Help Wound Healing Research? Real Peptides. 64.5% vs. 45.6% vs. 28.2% wound area reduction (rat model); LED combination study (12.5-fold cell viability, 230% bFGF, 70% collagen). https://www.realpeptides.co/does-ghk-cu-help-wound-healing-research/ ↩ ↩2
-
Food-Derived Tripeptide–Copper Self-Healing Hydrogel for Infected Wound Healing. Biomaterials Research (2024). DOI: 10.34133/bmr.0139. Diabetic wound model outcomes, antimicrobial activity. https://spj.science.org/doi/10.34133/bmr.0139 ↩
-
In situ photo-crosslinkable hyaluronic acid-based hydrogel embedded with GHK peptide nanofibers for bioactive wound healing. ScienceDirect (2023). Controlled-release nanofiber delivery in wound bed. https://www.sciencedirect.com/science/article/abs/pii/S1742706123006116 ↩
-
Effects of Topical Copper Tripeptide Complex on CO₂ Laser–Resurfaced Skin. Archives of Facial Plastic Surgery (2006). DOI: 10.1001/archfaci.8.4.252. Post-laser healing acceleration. https://www.liebertpub.com/abs/doi/10.1001/archfaci.8.4.252 ↩
-
Glycyl-L-histidyl-L-lysine-Cu²⁺ attenuates cigarette smoke-induced pulmonary emphysema and inflammation by reducing oxidative stress pathway. Frontiers in Molecular Biosciences (2022). PubMed ID 35936787. NF-κB and Nrf2/Keap1 dual-axis mechanism; protein-level cytokine outcomes. https://pubmed.ncbi.nlm.nih.gov/35936787/ ↩
-
The glycyl-l-histidyl-l-lysine-Cu²⁺ tripeptide complex attenuates lung inflammation and fibrosis in silicosis by targeting peroxiredoxin 6. Redox Biology / PMC (2024). PMC11228880. Biotinylated pull-down, SPR Kd = 2.81 × 10⁻⁵ M, proteome analysis methodology. https://pmc.ncbi.nlm.nih.gov/articles/PMC11228880/ ↩
-
The tri-peptide GHK-Cu complex ameliorates... PMC (2017). PMC5295439. Acute lung injury: alveolar infiltration, SOD, TNF-α, IL-6 outcomes. https://pmc.ncbi.nlm.nih.gov/articles/PMC5295439/ ↩
-
Postmenopausal Osteoporosis: Menopause Hormone Therapy and Selective Estrogen Receptor Modulators. PMC (2023). PMC10721581. Epidemiology, RANKL/estrogen axis, BMD loss rates. https://pmc.ncbi.nlm.nih.gov/articles/PMC10721581/ ↩
-
Bone and Muscle Support in Ageing Women with Life Wave X49™ Patch. Biomedical Research (AHK-Cu pilot). NTx biomarker findings; BMD changes in two participants. https://biomedres.us/fulltexts/BJSTR.MS.ID.007513.php ↩ ↩2
-
GHK-Cu and Reproductive Health: Mechanisms, Fertility Benefits. Inovi Fertility and Genetics Institute. Endometrial vascularization, uterine injury rodent data, granulosa cell oxidative stress in vitro. https://www.inovifertility.com/blog/ghk-cu-and-reproductive-health-mechanisms-fertility-benefits/ ↩ ↩2 ↩3
-
Peptides for Enhancing Anti-Müllerian Hormone Levels in Reproductive Health. Inovi Fertility and Genetics Institute. AMH biology, granulosa cell context. https://www.inovifertility.com/blog/peptides-for-enhancing-anti-mullerian-hormone-levels-in-reproductive-health/ ↩
-
Expression and Purification of Recombinant GHK Tripeptides Are Able to Protect against Acute Cardiotoxicity from Exposure to Waterborne-Copper in Zebrafish. PMC (2020). PMC7564529. 1:10 GHK:Cu ratio cardioprotection; zebrafish model methodology. https://pmc.ncbi.nlm.nih.gov/articles/PMC7564529/ ↩
-
Modulation of Gene Expression in Human Breast Cancer MCF7 and Prostate Cancer PC3 Cells by the Human Copper-Binding Peptide GHK-Cu. OBM Genetics (2021). MCF7 caspase upregulation; MYC target downregulation; ER+ breast cancer cell line. https://www.lidsen.com/journals/genetics/genetics-05-02-128 ↩
-
The Effect of the Human Plasma Molecule GHK-Cu on Stem Cell Actions and Expression of Relevant Genes. OBM Geriatrics (2018). MSC trophic factor secretion (HGF, EGF, FGF); stemness marker upregulation. https://www.lidsen.com/journals/geriatrics/geriatrics-02-03-009 ↩
-
Are We Ready to Measure Skin Permeation of Modern Antiaging GHK–Cu Tripeptide Encapsulated in Liposomes? PMC (2025). PMC11721469. Franz diffusion cell limitations; multi-layer measurement methodology; liposomal penetration improvement. https://pmc.ncbi.nlm.nih.gov/articles/PMC11721469/ ↩
-
GHK-Cu Dosing Protocol — Skin, Hair & Tissue Repair Guide (2026). Peptide Dosing Protocols. Subcutaneous half-life <60 min; safety profile; adverse event summary. https://www.peptidedosingprotocols.com/protocol/ghk-cu ↩ ↩2 ↩3
